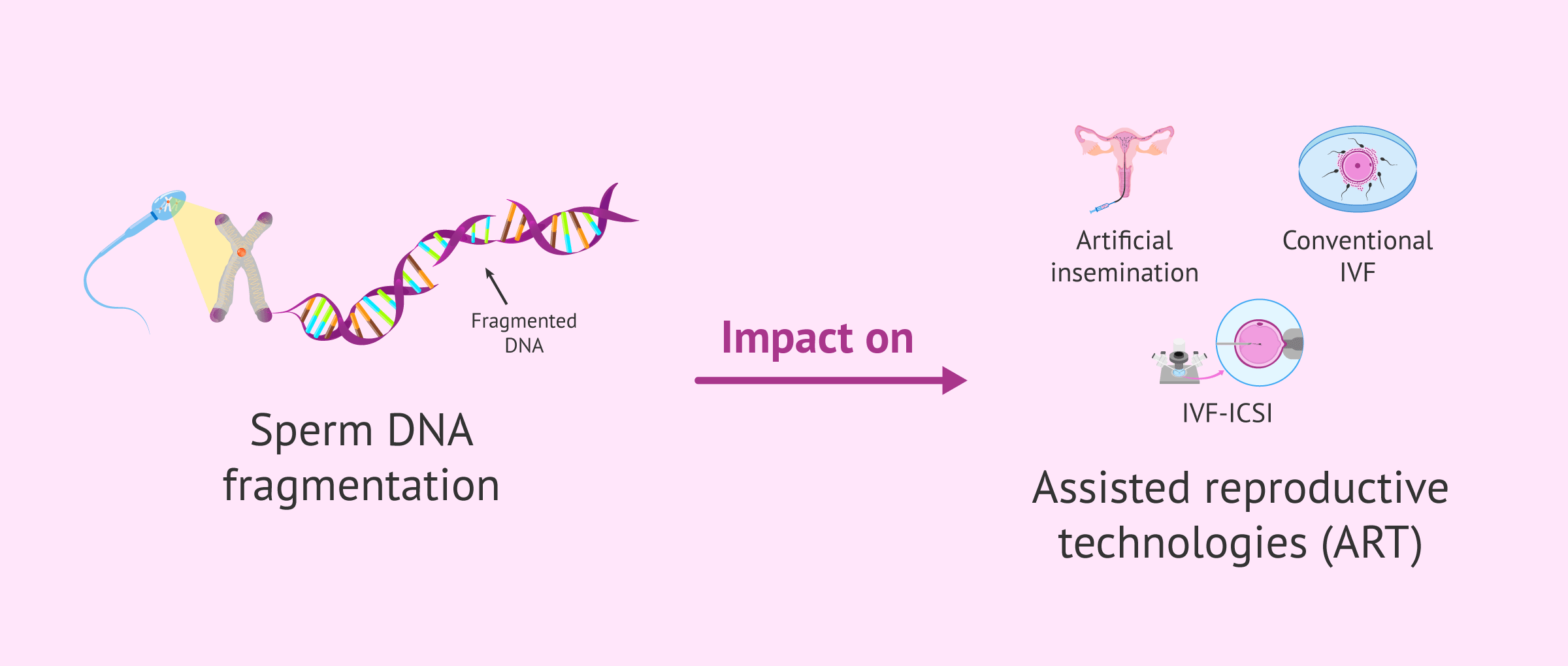
Semen quality has traditionally been evaluated using basic parameters such as sperm concentration, motility, and morphology. However, in recent years, the integrity of the sperm's genetic material has taken on a crucial role.
A recent study conducted by researchers from the Reproductive Medical Center, Hainan Women and Children’s Medical Center and the Department of Urology of the Haikou Affiliated Hospital of Central South University Xiangya School of Medicine, has shed new light on how sperm DNA damage affects different assisted reproduction techniques.
Provided below is an index with the 7 points we are going to expand on in this article.
What is DNA fragmentation?
Male infertility accounts for nearly half of the cases of reproductive problems in couples. Beyond conventional analyses, sperm DNA fragmentation (SDF) has established itself as a key biomarker of sperm quality. Basically, it refers to breaks or lesions in the DNA strand of the sperm.
This genetic integrity is fundamental not only for the fertilization of the egg to occur, but also for the subsequent development of the embryo and the maintenance of the pregnancy. When these levels of fragmentation in the sperm DNA are high, the chances of reproductive success can be compromised.
It is important to note that there are different laboratory tests to measure this sperm DNA damage, such as the SCSA, SCD, TUNEL, or Comet assays. The technique used may vary depending on the indication and the assisted reproduction center.
Impact on conventional in vitro fertilization
The research carried out by doctors Bangbei Wan, Yu Fu, Ning Ma, Zhi Zhou, and Weiying Lu, which consists of an "umbrella review" (a comprehensive analysis of previous meta-analyses), has evaluated more than 25,000 assisted reproduction cycles.
The results for conventional in vitro fertilization (IVF) were as follows:
- There is a significant association between high DNA fragmentation and a lower clinical pregnancy rate.
In conventional IVF, where the sperm must penetrate the egg on its own or with limited help, sperm DNA damage seems to act as a natural barrier that decreases the chances of successful fertilization.
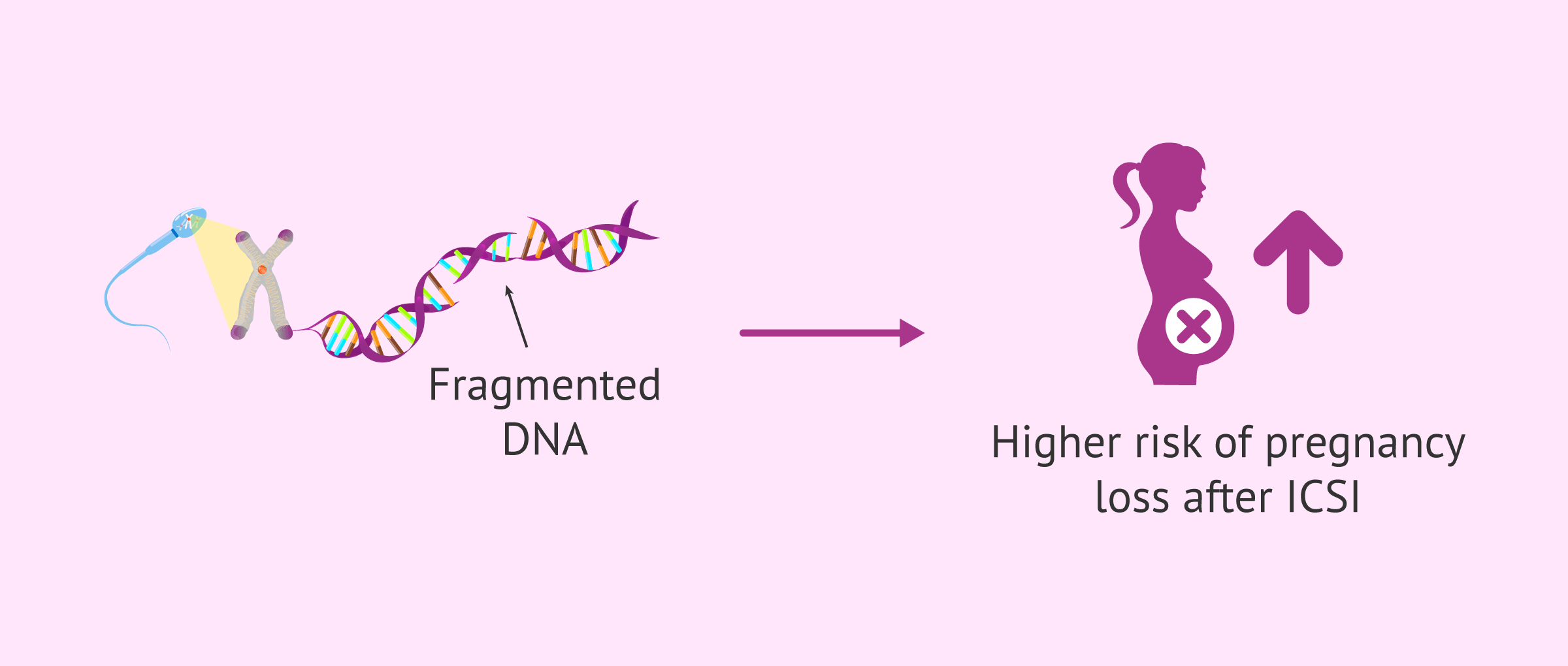
Risk of miscarriage in ICSI cycles
The case of intracytoplasmic sperm injection (ICSI) presents different and very important nuances for patients. In this technique, the embryologist selects the sperm and introduces it (by microinjection) directly into the egg, bypassing natural selection barriers.
According to the study, although the impact of sperm DNA fragmentation on the clinical pregnancy rate is weak after ICSI, the most striking data lies in pregnancy loss:
- A strong association was observed between high DNA fragmentation and a higher risk of miscarriage in ICSI cycles.
- By forcing fertilization with a sperm that could have damaged DNA, the embryo can be generated, but it has more difficulties evolving, which can lead to losses later on.
These findings suggest that, although ICSI helps achieve fertilization, the genetic damage brought by the male gamete can manifest in later stages of pregnancy.
Results in artificial insemination
For couples who opt for artificial insemination (AI), the quality of the sperm DNA is also decisive. The study concludes that high sperm DNA fragmentation is significantly associated with a reduction in clinical pregnancy rates.
Since insemination depends largely on the natural ability of the sperm to ascend and fertilize, extensive damage to its genetic load reduces the chances of success. Therefore, in cases of repeated insemination failures or unexplained infertility, evaluating fragmentation is a valuable diagnostic tool to guide treatment towards more complex options if necessary.
Community and Support
At inviTRA we work to make monthly and rigorous information accessible to everyone. If this article has helped you, consider supporting us so we can continue accompanying more people on their journey to parenthood.
References
Wan B, Fu Y, Ma N, Zhou Z, Lu W. Sperm DNA fragmentation and assisted reproduction: an umbrella meta-analysis. Eur J Med Res. 2026 Jan 7;31(1):218. doi: 10.1186/s40001-025-03801-y. PMID: 41501955; PMCID: PMC12870536. (View)