Freezing and vitrification are two different techniques used in assisted reproduction for the cryopreservation of eggs, sperm, and embryos. The objective of both processes is the same: to keep the cells intact for later use.
However, there are significant differences between the freezing method and vitrification, which determine the application of one technique or another depending on each case. Therefore, the cryopreservation of eggs and embryos is carried out through vitrification, while the cryopreservation of semen is done through slow freezing.
Provided below is an index with the 10 points we are going to expand on in this article.
- 1.
- 2.
- 2.1.
- 2.2.
- 3.
- 4.
- 5.
- 6.
- 6.1.
- 6.2.
- 6.3.
- 6.4.
- 6.5.
- 6.6.
- 7.
- 8.
- 9.
- 10.
Differences between cryopreservation and vitrification
When Assisted Reproductive Technology (ART) began to be popular, freezing or cryopreservation, also known as slow freezing, was the only technique available. Since this method offered high success rates in the case of sperm cryopreservation, today it continues to be the technique used for preserving sperm cells.
As regards oocytes and embryos, however, due to the larger size of their cells, their structure and functionality were compromised after the freezing-thawing process. Subsequently, the survival rates were too low, and this is the reason why, from that moment onwards, research focused on the pursuit of a method that allowed us to improve the success rates.
That is how the so-called vitrification method, also known as fast freezing, was born, which is the technique used nowadays for the cryopreservation of unfertilized eggs and embryos.
The birth of the vitrification method translated into a major step forward in the field of Assisted Reproduction, as it improved the survival rates of both oocytes and embryos, thereby avoiding the loss of quality caused after the thawing process.
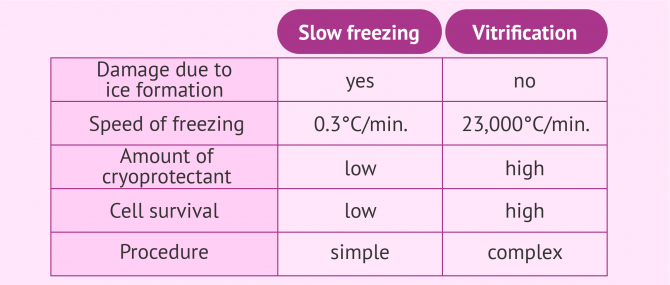
Vitrification consists in freezing or cryopreserving eggs and embryos with the minimum volume and ultra-rapidly. By doing this, we can prevent the formation of intracellular ice, which can damage the inner parts of the cells to a large extent.
With the vitrification technique, however, the concentration of cryoprotectant necessary for causing the dehydration of cells should be higher, which can be toxic. This is the reason why adjusting the exposure times as accurately as possible is crucial.
If the cells do not exceed the established contact time with the cryoprotective substances, vitrification offers important advantages. In the following image we can see a summary of the main differences between the two methods:
Egg vitrification
To proceed with egg vitrification, firstly it is necessary to replace the liquid contained within the oocytes with a substance called cryoprotectant, which role is to protect the inner cells of the unfertilized eggs from low temperatures.
Thus, to switch from the culture temperature (37°C) to the temperature necessary for cryopreserving the egg cells (-196°C), they go through different cryopreservation media that contain an increasing concentration of cryoprotectant.
The following is a diagram displaying the vitrification process step by step. To be precise, what you can see in the picture below is how the process works with the Cyrotop system, the most widely used nowadays. Nevertheless, there exist other methods, such as CryoTip, though its use is less common. In any case, the process followed is quite similar.
With Cryotop, or whatever the system of choice, the egg cells are preserved in tanks containing liquid nitrogen (N2) until they are necessary for use in future IVF cycles. That is the moment when they will be thawed.
Thawing is the opposite process. It involves switching from the cooling temperature to the culture temperature, and then putting them in different media that will allow us to modify their contents, thereby removing the concentration of cryoprotectant.
If you want to learn more about this process, we recommend that you visit the following post: Egg vitrification.
Classical cryopreservation or slow freezing reduced dramatically the quality of the oocytes, as the thawing process damaged the inner parts of the egg cell. Such damage reduced considerably the recovery potential, which made it very difficult for fertilization to occur and result in an embryo. This represents a huge improvement, as it allows for fertility preservation, and at the same time improves the accuracy of the egg donation process.
Female fertility preservation
The lifestyle and socio-economic situation of today's society have left many women no alternative but to delay their biological clock. Keeping in mind that a woman's pregnancy rate diminishes over time, egg vitrification translates into a major advantage for them. Thanks to the vitrification method, the eggs that were saved present almost the same quality as before vitrification. In other words, it is almost as if they have never been frozen.
Specifically, taking into account the data provided by the latest statistical record from the Spanish Society of Fertility (SEF) for the year 2023, the pregnancy rate using fresh own eggs was 34.6%, while with vitrified eggs it was 44.0%.
Also, this method allows a great number of cancer patients, women who need special treatment or medical intervention that could damage their eggs to become mothers.
Statistical data in this article shall not be used to imply or predict an outcome certainty to a specific individual within a population at risk.
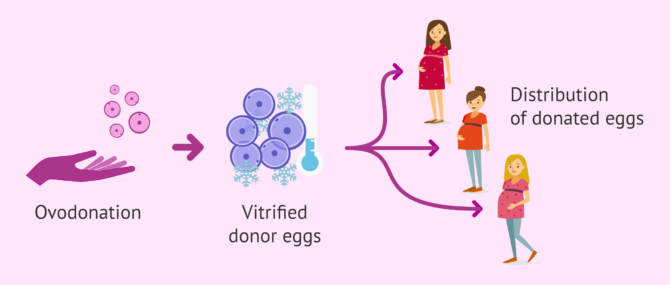
Egg donation
When the method used for cryopreservation was slow freezing, only fresh eggs could be used in donor-egg IVF cycles, as a great number of oocytes were at risk of being unusable after the thawing process. In addition, fresh donor cycles are not always feasible, as they require synchronization between the menstrual cycles of the egg donor and the recipient.
Egg vitrification has allowed us to optimize the egg donation process, offering, among other improvements, the chance to choose egg sharing as an alternative option. It involves using the oocytes from a single donor for the cycle of several recipients, which reduces the cost of the treatment, and allows us to make the most out of all the eggs collected from the same donor.
Embryo vitrification
The same applies with embryos since they are considered to be large cells as well. Thanks to ultra-rapid freezing or fast freezing, no damage is caused to their cell structures, which means that they continue growing and dividing with normality after being thawed.
The cryopreservation of embryos is a major advantage because it allows couples to store their unused, high-quality embryos after an IVF cycle for later use, without the need for the woman to undergo another ovarian stimulation cycle.
Moreover, vitrification is also a necessary procedure prior to embryo donation and adoption. This fertility treatment involves a couple who decides to donate their excess embryos after an IVF cycle to give another couple the chance to use them for starting a family. You have more information here: What is embryo donation and adoption?
The process of embryo vitrification works similarly to that followed for egg vitrification. However, if you want to learn more, do not miss this post: Embryo vitrification, how does it work?
Cryopreservation of sperm
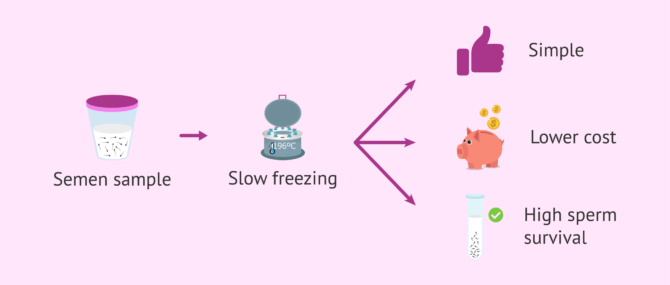
Since sperms are very small cells, their reduced volume prevents the intracellular formation of ice crystals, which could damage their structure.
This is the reason why contrary to what happens with embryos and egg cells, slow freezing procedures work well and continue to be used for freezing sperm samples.
Due to the simplicity of the procedure itself, and also because of its reduced cost, classical cryopreservation or slow freezing is still used when it comes to freezing sperm. The survival rates with this method are high, and their quality is almost as good as before being frozen.
Need more information? Read more here: Cryopreservation of sperm.
Costs and financing options
More often than not, fertility clinics do not include the cost of gamete and/or embryo cryopreservation and vitrification procedures when they give patients an initial quotation for IVF. For this reason, you are recommended to get as much information as possible beforehand regarding the approximate costs of these techniques.
If you are considering cryopreserving your gametes or embryos to have a baby in the future, we recommend that you start by getting a Fertility Report. In 3 simple steps, it will show you a list of clinics that fit your preferences and meet our strict quality criteria. Moreover, you will receive a report via email with useful tips to visit a fertility clinic for the first time.
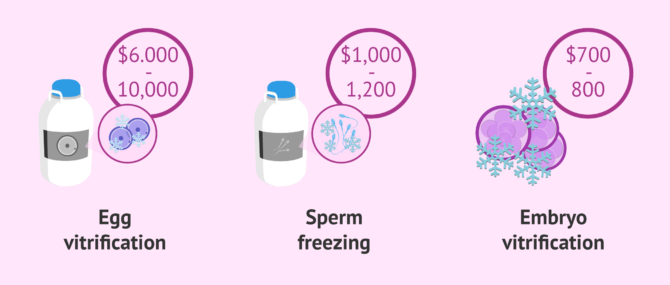
Even though fees vary per clinic and process followed for collecting and storing the samples, the simplest and cheapest option is sperm cryopreservation, which price ranges from $1,000 to $1,200. The breakdown of these fees includes the first appointment, blood draw, and an approximate annual storage fee of $250-450.
On the other hand, both egg and embryo vitrification are more complex techniques than sperm freezing, and subsequently more expensive. Thus, while egg vitrification can cost about $6,000-10,000—plus a storage fee of $800 per year approximately—, the cost of embryo vitrification services is $700-800 plus a storage fee per year of $250.
If you would like to know more about the costs of assisted reproduction techniques, you can visit this link: What is the price of assisted reproduction treatments?
FAQs from users
Is it true that it is free to freeze eggs for cancer patients?
There are some assisted reproduction centers that offer egg vitrification treatment to preserve fertility in oncology patients completely free of charge. However, this is not widespread.
Does using frozen semen affect the result of assisted reproduction treatment?
If we talk about whether the use of a frozen semen sample will affect the results, it will depend on what treatment is going to be carried out and what is the reason why it is decided to use a previously frozen sample and not a fresh sample.
It has been observed that freezing affects, to a greater or lesser extent, the mobility of the sperm, so in cases of severe male factor, the recommendation would be to use fresh semen.
In addition, donor semen samples are frozen (Spanish law does not allow the use of a fresh donor), so the use of frozen semen does not have to worsen the results except in certain particular cases.
On the other hand, the use of frozen samples allows us to have more flexibility when organizing treatments both for the needs of our patients and for the management of daily work in our clinics.
Which is better? Egg freezing or egg vitrification?
Without a doubt, vitrification. This technique is the one that has really made it possible to offer the technique of female preservation with all the guarantees.
First of all it would be necessary to distinguish the two techniques and their history, as well as why the conventional freezing technique was relatively valid for embryos but not for oocytes.
With the conventional freezing technique, cryoprotectants are used in low concentration for a relatively long time and a slow progressive cooling. This sometimes results in the formation of crystals that are lethal to the cells. The larger the cell, the greater the possibility of crystals forming, and if we take into account that the egg is the largest cell in the human body, this explains why this technique has produced very poor results with eggs.
The vitrification technique consists of exposing embryos and oocytes to high concentrations of cryoprotectant in a relatively short time and ultra-fast cooling (in the order of 20,000 ºC/min). As a result of this process a vitreous mass is formed (hence the name vitrification) avoiding the formation of crystals that would damage the cell.
Therefore, vitrification of eggs is undoubtedly better than freezing. It is this technique that has really made it possible to offer the technique of female preservation in a standardized way with all the guarantees.
Can I still freeze my eggs at 40?
Egg quality diminishes as the woman ages. Such decrease is more apparent from age 35, and that is the reason why the general recommendation is to cryopreserve them before this age. By doing this, their quality will be higher, and subsequently their survival rates, and the chances of getting pregnant in the future.
What is the cost of egg freezing?
The price of egg freezing using the vitrification technique usually varies from one clinic to another. In any case, the cost is usually between 5.200$ and 6.000$.
Is special treatment required for egg vitrification?
Yes, in order to vitrify the eggs, it is necessary to extract them through follicular puncture. In addition, in order to obtain a greater number of eggs in this surgical intervention, the patient will follow a controlled ovarian stimulation treatment.
Therefore, the woman who is going to vitrify her eggs, either to preserve fertility, to undergo an IVF process or for egg donation, will have to go through these two processes: medical stimulation of ovulation and ovarian puncture.
Recommended readings
If you are reading this because you are considering preserving your fertility, you might be interested in visiting the following article: Fertility preservation: cost and options.
Also, in cases of egg donation, one can choose between undergoing a fresh or a frozen cycle, as explained above. Wondering whether there is a huge difference between these two options? Clear your doubts here: Frozen donor eggs vs. fresh donor eggs.
Community and Support
At inviTRA we work to make monthly and rigorous information accessible to everyone. If this article has helped you, consider supporting us so we can continue accompanying more people on their journey to parenthood.
References
ESHRE Task Force on Ethics and Law. Oocyte criopreservation for age related fertility loss. Human. Reprod. 2012. 27:1231-1237 (View)
Fertility Assessment and treatment for people with fertility problems. NHS. NICE clinical guideline 156. February 2013 (View)
Alberto Reche y Manuel Martínez Moya. Preservación de la fertilidad. Manual de Buena Práctica Clínica en Reproducción Asistida (pp. 115-131). Sociedad Española de la Fertilidad.
Glujovsky D, Riestra B, Sueldo C, Fiszbajn G, Repping S, Nodar F, Papier S, Ciapponi A. Vitrification versus slow freezing for women undergoing oocyte cryopreservation. Cochrane Database of Systematic Review 2014, Issue 9. Art. No.: CD010047 (View)
Sociedad Española de Fertilidad. Registro Nacional de Actividad 2023-Registro SEF. (View)
FAQs from users: 'Is it true that it is free to freeze eggs for cancer patients?', 'Does using frozen semen affect the result of assisted reproduction treatment?', 'Which is better? Egg freezing or egg vitrification?', 'Can I still freeze my eggs at 40?', 'Can you freeze sperm at home for later use?', 'Freezing sperm and getting a vasectomy, is it possible?', 'What is the cost of egg freezing?' and 'Is special treatment required for egg vitrification?'.
Authors and contributors
More information about Cristina Algarra Goosman








Hello, is sperm vitrification also possible? I mean, I know it’s not widely used, but does it exist, too?
Dear Martha,
Sperm cryopreservation is done using classical freezing procedures, as they guarantee high survival and success rates after thawing the sample. However, it is true that researchers are trying to improve sperm cryopreservation techniques and the possibility of vitrifying sperm with good outcomes also exists. For the moment, it’s just an experimental procedure which still needs further investigation.
I hope this helps,
Best
I’d like to know if it’s possible to ship eggs from one clinic to another. I know it’s allowed with embryos, but not sure with eggs.. Thank you.
Dear lucy,
Yes, it is possible to ship eggs as well as embryos, but depending on the country the conditions at which it can be done may vary. Ask your clinic for further information to make sure everything is done as it should.
Best