Fragile X syndrome (FXS), also known as Martin-Bell syndrome, is a genetic disorder that manifests mainly through mental retardation and developmental problems martin-Bell syndrome fragile X syndrome (FXS), is a genetic disorder that manifests itself mainly through mental retardation and developmental problems.
The diagnosis of this syndrome consists of detecting the presence of a mutation in the FMR-1 gene, responsible for the onset of this disease. This gene is located on the X sex chromosome, which is severely affected by the presence of this mutation.
Fragile X syndrome is the most common form of hereditary moderate mental retardation, second only to Down syndrome.
Provided below is an index with the 11 points we are going to expand on in this article.
- 1.
- 1.1.
- 2.
- 2.1.
- 2.2.
- 3.
- 4.
- 5.
- 6.
- 7.
- 7.1.
- 7.2.
- 7.3.
- 7.4.
- 7.5.
- 7.6.
- 8.
- 9.
- 10.
- 11.
What is the Fragile X Syndrome?
Fragile X syndrome is an X-linked genetic disease caused by a mutation in the FMR-1gene. It is a change in the DNA sequence that causes the absence of a protein called FMRP1 (Fragile X Mental Retardation Protein). This protein is essential for normal brain development, which explains the problems in the development of people affected by this syndrome.
The name of the syndrome is due to the fact that this mutation causes a small break in one of the arms of the X chromosome known as the fragile site. This breakage is only apparent when the chromosome is treated under specific conditions in the laboratory.
The prevalence of Fragile X syndrome is 1 in 4,000 males and 1 in 6,000 females worldwide. Therefore, this syndrome is one of the most frequent rare diseases in humans.
Causes
This genetic disease is caused by a mutation in the FMR-1 gene sequence. This change causes the protein for which this gene codes(FMRP1) is not produced and does not perform its function.
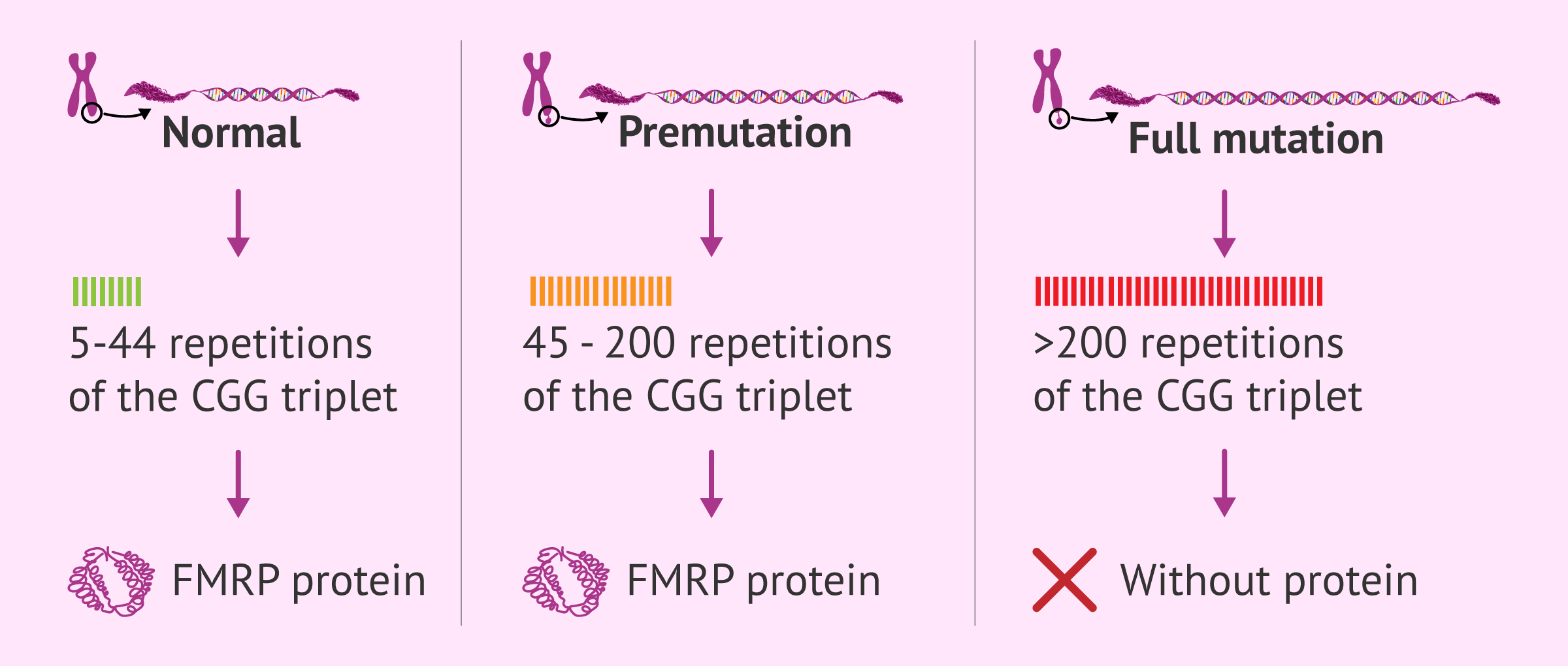
Within the FMR-1 gene there is a specific segment characterized by the repetition of a specific genetic sequence: the CGG trinucleotide. This sequence is present between 5 and 40 times in people not affected by Fragile X syndrome.
However, if the number of repeats increases, the genetic code of this gene is altered. As a consequence, a series of molecular mechanisms are initiated that completely inactivate the gene, as well as the production of its corresponding protein.
The severity with which this disease affects the person with the mutation depends on the number of expansions of the CGG sequence. According to this, the mutation holder will be:
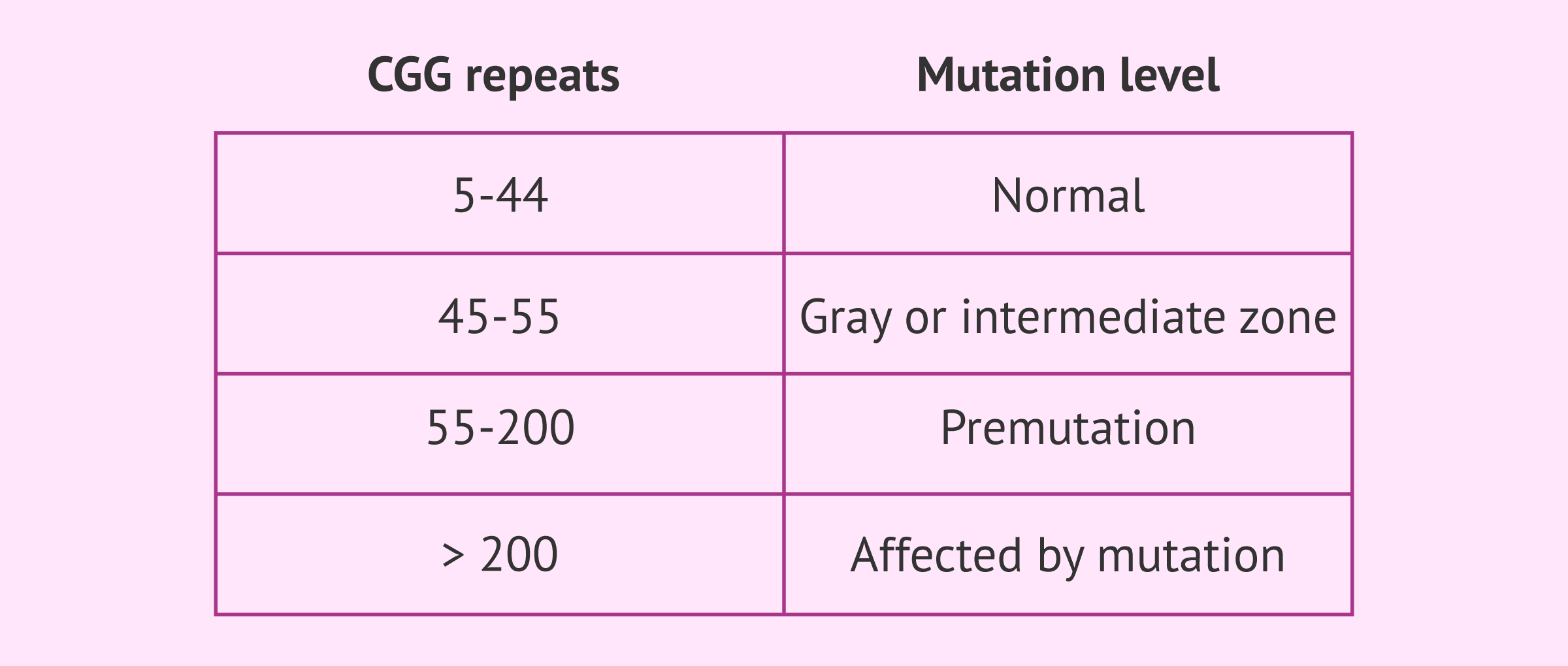
- Carrier: when the CGG sequence is repeated between 45 and 200 times. This condition is called premutation and the person who has it does not develop the disease, because the functioning of the gene is not so affected. However, it is capable of transmitting it to its offspring, with the consequent risk that the offspring will become ill. It is worth noting that between 45 and 55 repeats of the CGG triplet is known as the gray zone o intermediate zone and individuals do not have the syndrome although they may have some symptoms, but from 55 onwards they do acquire the premutation level and become carriers of the disease.
- Affected: the diseased person possesses the repeat in a number greater than 200, which is considered a complete mutation. In this case, symptoms do develop to a greater or lesser degree.
Although rare, cases have also been reported of patients with Fragile X syndrome who had point mutations or deletions of the FMR1 gene, but still produced a protein deficiency.
Inheritance of Fragile X syndrome
The inheritance of the mutation that causes this syndrome is dominant, i.e. only one of the two copies of the FMR-1 gene being mutated, the disease can develop depending on the number of repeats of the CGG triplet.
However, there is an added peculiarity to this pathology, since the transmission of Fragile X syndrome is linked to the X chromosome. This expression is due to the fact that the mutated gene is located on the X sex chromosome and, therefore, implies that its appearance and degree of affection is different in each sex, since men have only one X chromosome and women have two.
Fragile X syndrome in males
Since males have only one X chromosome in their karyotype, if they have inherited the complete mutation, the disease will occur.
This means that this disease is mainly suffered by men and the symptoms they suffer are more severe than in the case of women.
Fragile X syndrome in women
Females, on the other hand, are characterized by having 2 sex chromosomes X. Despite this, not all genes on both chromosomes are expressed, as this would cause them to express twice as many genes as males. To avoid this, one of the two chromosomes is randomly deactivated in each cell of the woman's body.
This is a great advantage, because if the deactivated chromosome happens to be the one with the mutation, protein production would be completely normal because the unaffected chromosome compensates for it. However, the female will be able to transmit the mutation to her offspring because she has the altered copy.
However, if the deactivated chromosome is the healthy one, the mutated gene will remain active and the production of the protein will be altered, contributing to the development of the disease.
Therefore, the symptoms and the degree to which this syndrome affects women are much milder than in the case of men. There are fewer cases of women with this disease, as they are mainly carriers.
Disease transmission
Another important feature to take into account in the transmission of this disease is that the passing of the mutation from one generation to another favors an increase in the number of repetitions and, therefore, the risk of developing the syndrome is also higher.
This depends to a large extent on the size of the permutation that the individual carrier possesses since it has been scientifically proven that the greater the number of repetitions, the greater the predisposition to further increase them and to develop the disease.
Physical characteristics and symptoms
Although the physical and clinical features of this disease are very specific, it is important to always perform a genetic diagnosis to clarify the presence of the mutation in chromosome X.
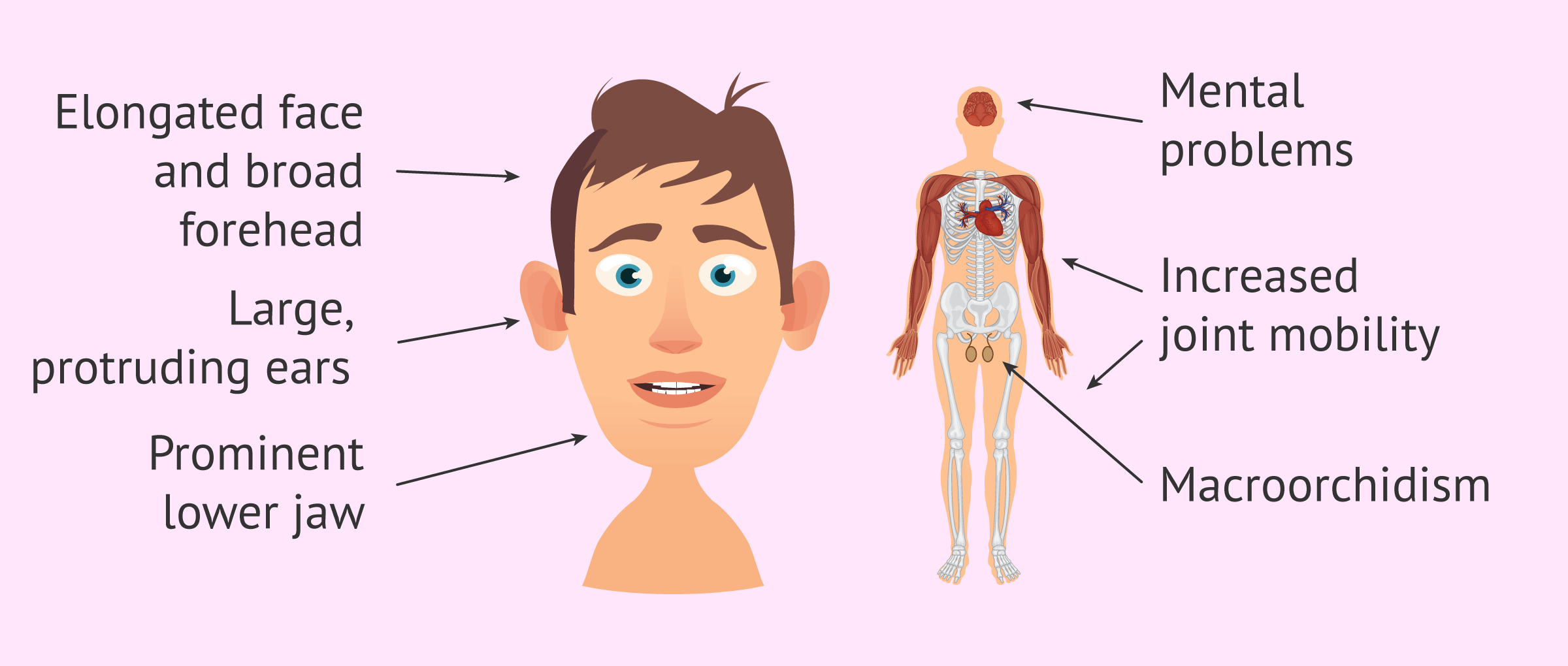
The most frequent clinical manifestations of Fragile X syndrome are the following:
- Coarse facial features.
- Elongated face with broad forehead.
- Large and detached ears.
- Increased joint mobility.
- Macroorchidism or increase in testicular size.
- Learning disabilities.
Other symptoms that may also occur are macrocephaly, mouth anomalies, flat feet, bowed knees, and other ophthalmologic, cardiac, and/or endocrine alterations.
However, the most important presenting symptom in this disease is mental retardation and neurological defects.
Fragile X syndrome is included in the so-called autism spectrum and it is common for patients with this syndrome to present hyperactivity, attention deficit or aggressive behavior, among others. Developmental disorders are another symptom that can occur at the motor and language levels. Likewise, sensory integration disorders, sleep disorders, epilepsy, and even some irregularities in the brain may appear.
The explanation for these alterations is given by the function of the protein that is altered in this disease. Although its function is not yet fully understood, it is known that it plays a very important role in the transmission of nerve impulses. This is why the lack of this protein causes the most pronounced damage to the central nervous system.
Treatment of Fragile X
Since Fragile X syndrome is caused by the absence of the FMRP protein, the optimal intervention would be to restore the function of this protein. Currently, there are several lines of research in this regard, although there is no treatment for Fragile X syndrome.
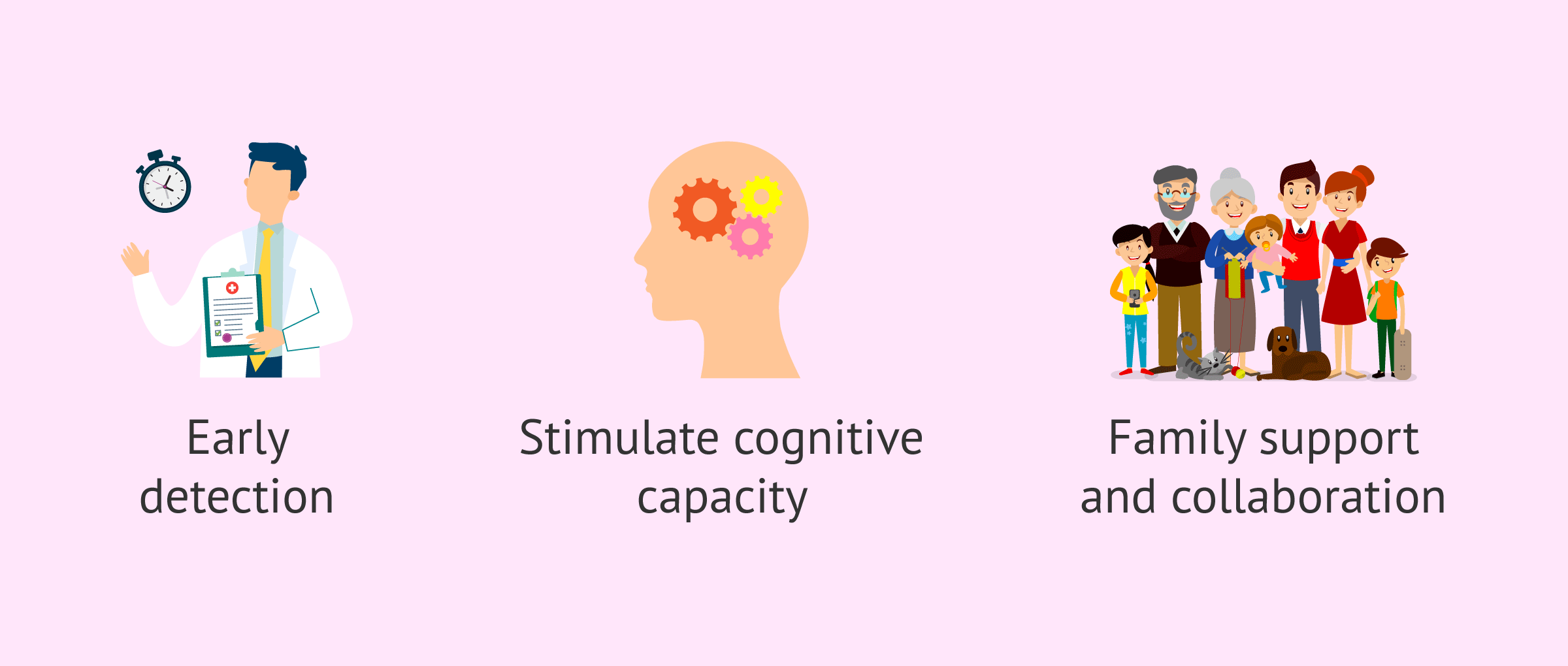
Given the irreversibility of this condition, current treatment is focused on stimulating the cognitive abilities of the affected person.
It has been proven that the earlier these types of educational therapies are implemented, the more productive the results will be. For this, early detection of the disease is important, as well as the support and collaboration of the family.
These types of pedagogical interventions are often combined with pharmacological treatment, especially to mitigate hyperactivity, anxiety or attention deficit.
Fragile X syndrome and fertility
This disease also affects the reproductive health of carriers and patients. Women carrying the premutation have an increased risk of developing early ovarian failure. In addition, these women have a lower ovarian reserve that can lead to the development of juvenile menopause. Approximately 20% of Fragile X carriers suffer from this disorder.
In the event of premature depletion of the ovarian reserve, the most common reproductive option available to these women is to resort to oocyte donation treatment.
If it is the man who suffers from Fragile X Syndrome, donor sperm can also be used to prevent the transmission of the pathology to the offspring.
These reproductive alternatives are very effective, as they totally prevent children from being sick. Egg and/or sperm donors are specially screened for the presence of this genetic alteration.
Assisted procreation, as any other medical treatment, requires that you rely on the professionalism of the doctors and staff of the clinic you choose. Obviously, each clinic is different. Get now your Fertility Report, which will select several clinics for you out of the pool of clinics that meet our strict quality criteria. Moreover, it will offer you a comparison between the fees and conditions each clinic offers in order for you to make a well informed choice.
Finally, it is also possible to perform embryo selection through the preimplantation genetic diagnosis (PGD) process. Thanks to this technique it is possible to genetically analyze the embryos to determine those that do not have the mutated gene and select them for transfer to the woman's uterus. This avoids the need to resort to donated gametes.
If you want more information about this reproductive option, you can click on this link: Preimplantation genetic diagnosis.
FAQs from users
What is the relationship between fragile X and early menopause?
Fragile X syndrome is a genetic disease characterized by the expansion of a region of the X chromosome. This expansion causes a certain region of the X chromosome to repeat over and over again. An excess number of repeats will determine what degree of the disease is present. In this phenomenon we can find three situations: the non-mutated, the mutated and the pre-mutated.
The non-mutated comprise the majority of the population. The number of repeats in this region is low and no problem or disease develops.
The mutated are those patients who will have a much higher number of repeats than the non-mutated and pre-mutated individuals, which can lead to the development of disease.
Those who are pre-mutated will have a high number of repeats but will not manifest the disease. However, these patients are at risk of having an increased number of repeats in their offspring and may develop the disease.
This group of women usually have a low ovarian reserve at an early age, which can lead to early menopause. That is why when young women with low ovarian reserve come to the clinic it is necessary to perform a test to determine that they do not have a fragile X pre-mutation.
Read more
What treatment is advised in a Fragile X syndrome situation?
Once Fragile X syndrome has been diagnosed and in order to avoid having an affected child, the treatment to achieve pregnancy should be in vitro fertilization (IVF) with preimplantation genetic diagnosis (PGD) in cases of good ovarian reserve or oocyte donation in cases of low ovarian reserve and ovarian failure.
Read more
How is the molecular diagnosis of Fragile X Syndrome performed?
Briefly, it consists of detecting the amplification of the CGG repeat in DNA samples obtained from peripheral blood using a test called PCR (Polymerase Chain Reaction).
This test is the most reliable method that exists to determine if the individual is a carrier of the premutation or the full mutation and what risk they have of transmitting it to their offspring.
What is the life expectancy of those affected by this disease?
Fortunately, this syndrome does not affect the longevity of the people who suffer from it, since it has been proven that they have an average life span similar to that of the non-diseased population.
Is it possible to detect Fragile X in prenatal diagnosis?
Yes, through tests such as amniocentesis it is possible to predict, before birth, whether or not the fetus will have the disease.
Is there any cure or way to prevent this syndrome?
Once diagnosed, the syndrome cannot be prevented, as it is in the individual's genes and these cannot be modified. Furthermore, there is no treatment for Fragile X syndrome.
In these situations, treatment is focused on improving the affected person's abilities, whether it is learning to walk, to control behavior, etc.
Recommended readings
Although it is not one of the most characteristic symptoms, it is possible that certain alterations may occur in the fertility of those affected by this disease. In this article, we have very briefly mentioned this situation and the possible reproductive options. For more information on treatments, please visit the following article: Assisted reproduction techniques: differences and complexity.
As we have already mentioned, one of the possible conditions derived from this alteration in carrier women is early menopause. We tell you what it is in more detail in this article: What is early menopause?
Community and Support
At inviTRA we work to make monthly and rigorous information accessible to everyone. If this article has helped you, consider supporting us so we can continue accompanying more people on their journey to parenthood.
References
Coffman MC, Shaffer RC Schmitt LM, Dominick KC, Pedapati E, Wang A, Berry-Kravis E, Tartaglia N, Erickson CA. Examination of Correlates to Health-Related Quality of Life in Individuals with Fragile X Syndrome. Brain Sci. 2020 Apr 4;10(4). pii: E213. doi: 10.3390/brainsci10040213 (View)
De Vries BB, Halley DJ, Oostra BA, Niermeijer MF. The fragile X syndrome. J Med Genet. 1998;35:579-89 (View)
Hilvert E, Hoover J, Sterling A, Schroeder S. Comparing Tense and Agreement Productivity in Boys With Fragile X Syndrome, Children With Developmental Language Disorder, and Children With Typical Development. J Speech Lang Hear Res. 2020 Apr 13:1-14. doi: 10.1044/2019_JSLHR-19-00022 (View)
Mingarro Castillo MM, Ejarque Doménech I, García Moreno A, Aras Portilla LM. Síndrome del cromosoma X frágil. Rev Clin Med Fam vol.10 no.1 Albacete feb. 2017 (View)
Reiss AL, Hall SS. Fragile X syndrome: assessment and treatment implications. Child Adolesc Psychiatr Clin N Am. 2007;16:663-75 (View)
Rodrigues VO, Polisseni F, Pannain GD, Carvalho MAG. Genetics in human reproduction. JBRA Assist Reprod. 2020 Apr 15. doi: 10.5935/1518-0557.20200007 (View)
Sauna-Aho O, Bjelogrlic-Laakso N, Rautava P, Arvio M. Ageing and cognition in men with fragile X syndrome. J Appl Res Intellect Disabil. 2020 Apr 12. doi: 10.1111/jar.12733 (View)
Van Dijck A, Barbosa S, Bermudez-Martin P, Khalfallah O, Gilet C, Martinuzzi E, Elinck E, Kooy RF, Glaichenhaus N, Davidovic L. Reduced serum levels of pro-inflammatory chemokines in fragile X syndrome. BMC Neurol. 2020 Apr 15;20(1):138. doi: 10.1186/s12883-020-01715-2 (View)
FAQs from users: 'What is the relationship between fragile X and early menopause?', 'What treatment is advised in a Fragile X syndrome situation?', 'How is the molecular diagnosis of Fragile X Syndrome performed?', 'What is the life expectancy of those affected by this disease?', 'Is it possible to detect Fragile X in prenatal diagnosis?' and 'Is there any cure or way to prevent this syndrome?'.
Authors and contributors


More information about Cristina Algarra Goosman
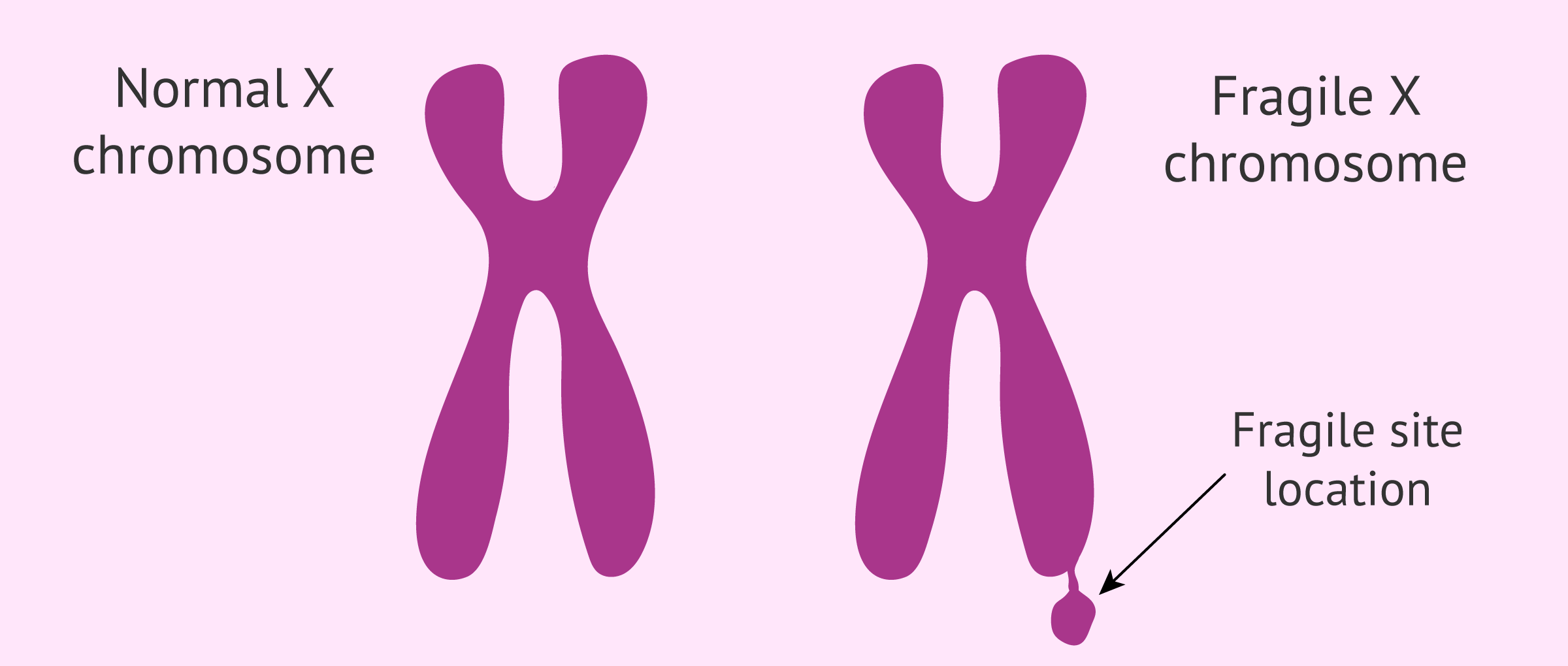
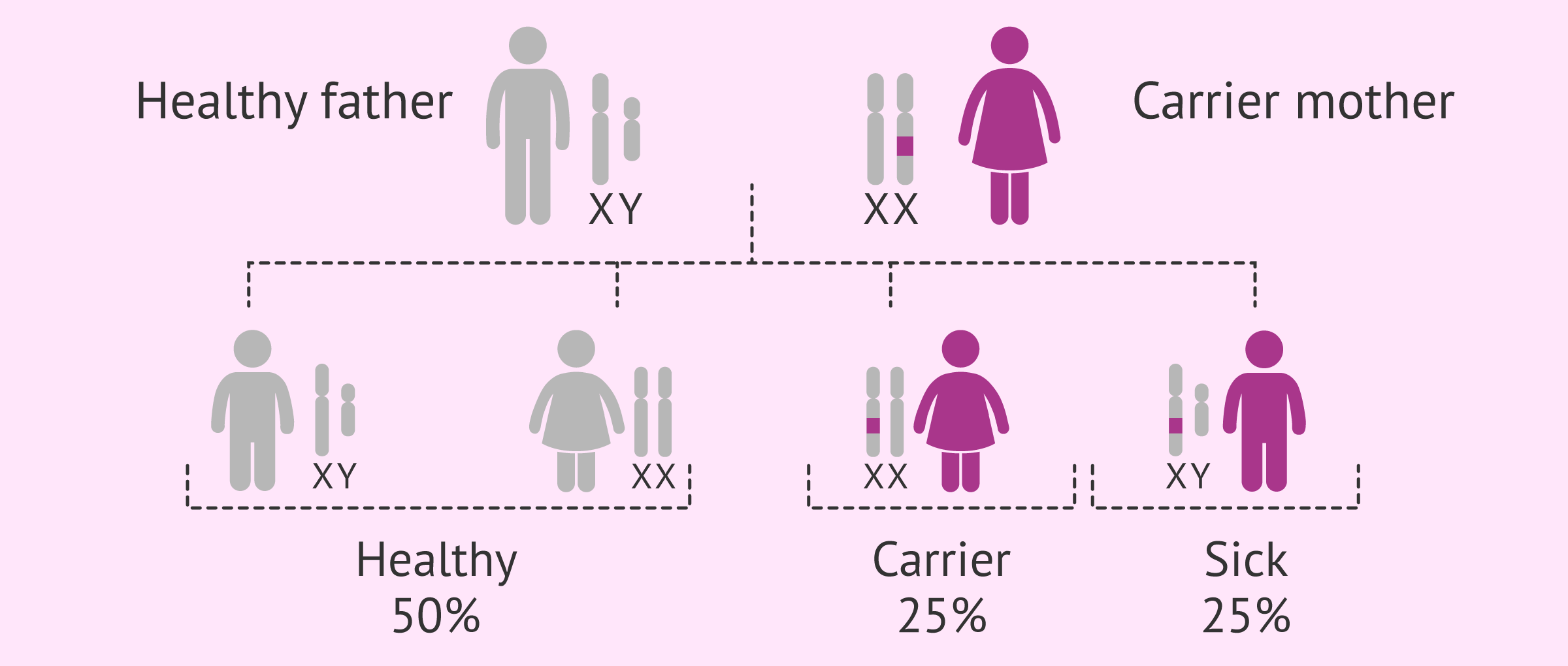




Hello, so from what I understand women cannot suffer from this syndrome?
Hi Emily,
This syndrome is associated with the X chromosome, so the male who suffers from it will have inherited the X chromosome mutation and therefore will present this disease.
Females on the other hand must have the mutation transmitted from both the father and the mother. This is complicated because generally, a male affected by Fragile X syndrome resorts to sperm donation in order not to pass the disease to his offspring.
For all these reasons, there is a higher prevalence of the disease in males than in females.
I hope I have answered your question.
Best regards
Hello, I am a carrier of this disease and I would like to know what I can do to avoid transmitting it to my children, thank you.
Hi Kelsey,
The transmission of this disease is associated with the X chromosome, so if you are a carrier, there is a 25% of chance of you having a daughter that is a carrier, a 25% of chance that you have a boy and he is affected by this disease, and a 50% chance of your offspring being healthy independent of being boy or girl.
Therefore, to prevent your offspring from being affected by this disease, a PGD of the embryos can be performed to see if they are affected or not by the disease.
I hope I have helped you.
Best regards.