Decapeptyl (triptorelin) is a gonadotropin-releasing hormone (GnRH) agonist analogue drug.
It is used in assisted reproduction treatments to control ovarian function in in vitro fertilization (IVF) cycles.
Provided below is an index with the 11 points we are going to expand on in this article.
- 1.
- 2.
- 3.
- 4.
- 4.1.
- 4.2.
- 5.
- 6.
- 7.
- 7.1.
- 7.2.
- 7.3.
- 8.
- 9.
- 10.
- 11.
Composition and Mechanism of action
The active principle of Decapeptyl is triptorelin and its excipients are: mannitol in the vial and sodium chloride and water for preparations to be injected into the vial.
Tryptorelin is an agonist analogue of GnRH. The main function of GnRH in the body is to promote or increase the release of follicle-stimulating hormone (FSH) and luteinizing hormone (LH), which are the gonadotropins involved in follicular development.
In women undergoing IVF treatment, the administration of this drug initially induces gonadotropic stimulation (FSH and LH). Once this first phase has passed, due to a decrease in sensitivity to this medication, the release of these hormones is inhibited.
The role of tryptorelin is to allow control of the function of the ovaries in ovarian stimulation cycles. It inhibits the LH peak that causes ovulation and thus prevents spontaneous ovulation. If this were to occur, it would cause the cycle to be cancelled because it would not be possible to obtain the eggs for fertilization in the laboratory.
In addition, it also allows monitoring of ovarian activity in patients with ovarian function who undergo frozen embryo transfers or egg donation cycles. This allows the endometrium to be in optimal conditions for embryo implantation.
Package and price
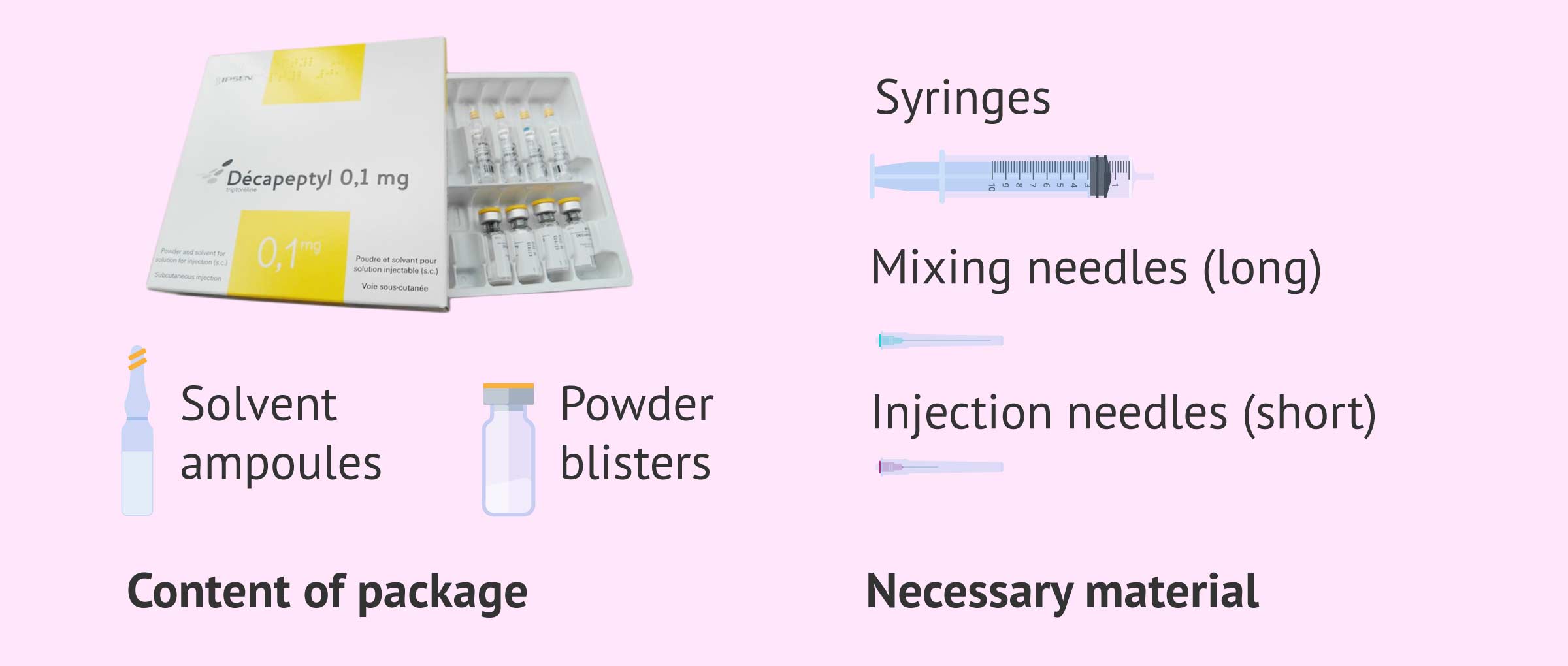
Decapeptyl can be found in four different presentations:
- Daily Decapeptyl
- contains 7 vials each containing 0.1 mg of triptorelin powder (acetate) and 7 vials of solvent. By reconstituting the drug, an injectable suspension is achieved.
- Monthly Decapeptyl
- contains 7 vials each containing 3.75 mg of triptorelin powder (acetate) and 7 vials of solvent. After reconstitution, an injectable suspension is obtained.
- Quarterly Decapeptyl
- contains 7 vials each containing 11.25 mg of triptorelin powder (acetate) and 7 vials of solvent. As in the previous presentations, by reconstituting the drug, an injectable suspension is obtained.
- Semiannual Decapeptyl
- contains 7 vials each containing 22.5 mg of triptorelin powder (acetate) and 7 vials of solvent. After reconstitution, in this case an extended release injectable suspension is achieved.
In assisted reproduction treatments, both daily and monthly Decapeptyl are used. Depending on which protocol is carried out, one or the other will be administered.
Indications
Decapeptyl is indicated to treat female sterility, as a complementary treatment to gonadotropins (HMG, FSH, hCG) during ovulation stimulation in assisted reproduction treatments.
It is also used in patients with ovarian function who undergo embryo transfer in egg donation or frozen embryo cycles.
Monthly Decapeptyl is also indicated in other pathologies, such as for the treatment of endometriosis, uterine fibroids, early puberty and prostate cancer.
However, quarterly and semi-annual presentations are only indicated in patients with prostate cancer.
How is Decapeptyl administered?
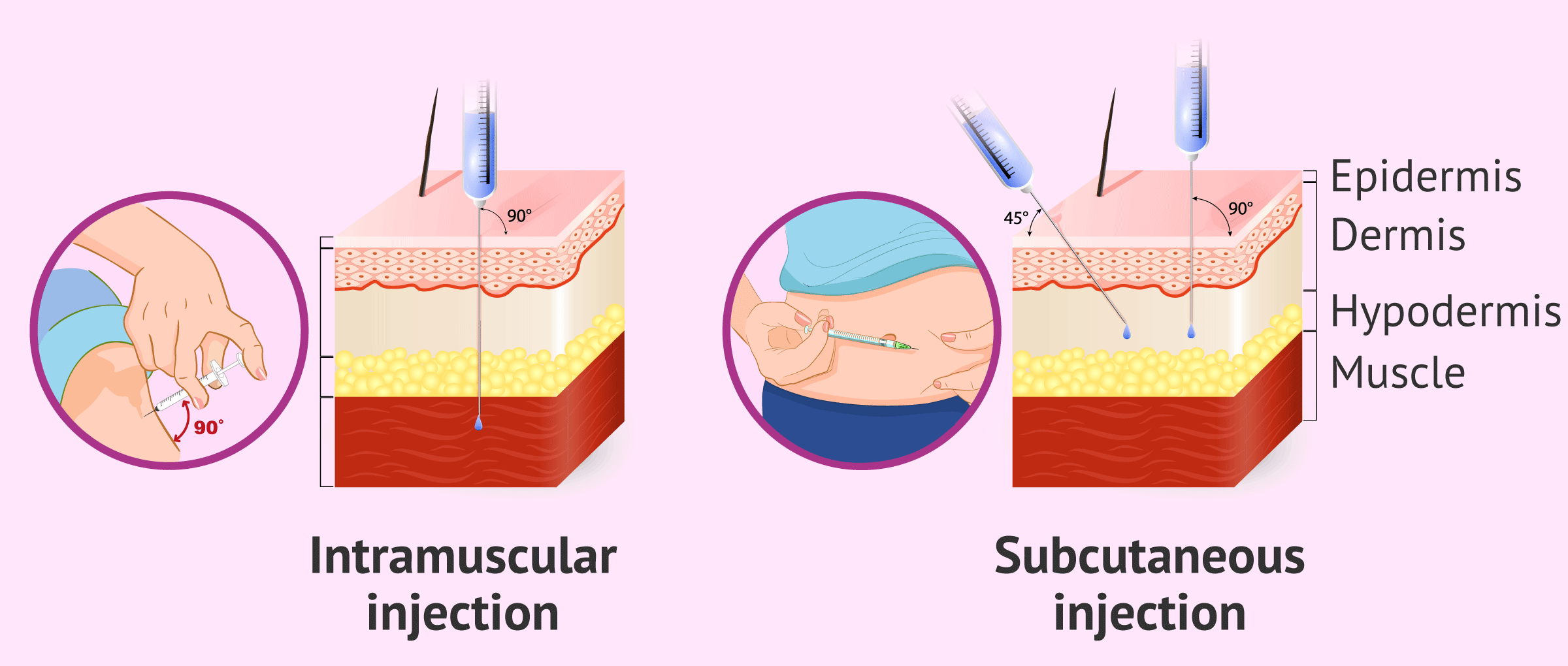
The route of administration of daily Decapeptyl is subcutaneous, in the tissue located immediately under the skin. The rest of the presentations require intramuscular administration, directly into the muscle.
The main difference between both types of administration is the depth at which the injection is performed, being deeper the intramuscular one, which is usually performed in the buttocks or in the arm. The needle should enter at a 90º angle. On the other hand, in the case of subcutaneous injection, it can be done from 45º to 90º.
Because intramuscular injection is recommended to be performed by health care personnel, this article will focus on the subcutaneous administration of daily Decapeptyl, which is usually self-injected into the abdominal area.
For the injection, in addition to the drug, it will be necessary to have a syringe, an intramuscular injection needle, a subcutaneous injection needle, gauze and alcohol.
Drug Reconstitution
The first step will be to reconstitute the drug before injecting it. To do this, the following steps must be taken:
- Remove the plastic cap from the glass vial where the powder is.
- Gently tap the top of the solvent vial to move the liquid to the bottom of the vial.
- Place the vial with the blue dot facing you, cover the top with gauze so you don't cut yourself and break it.
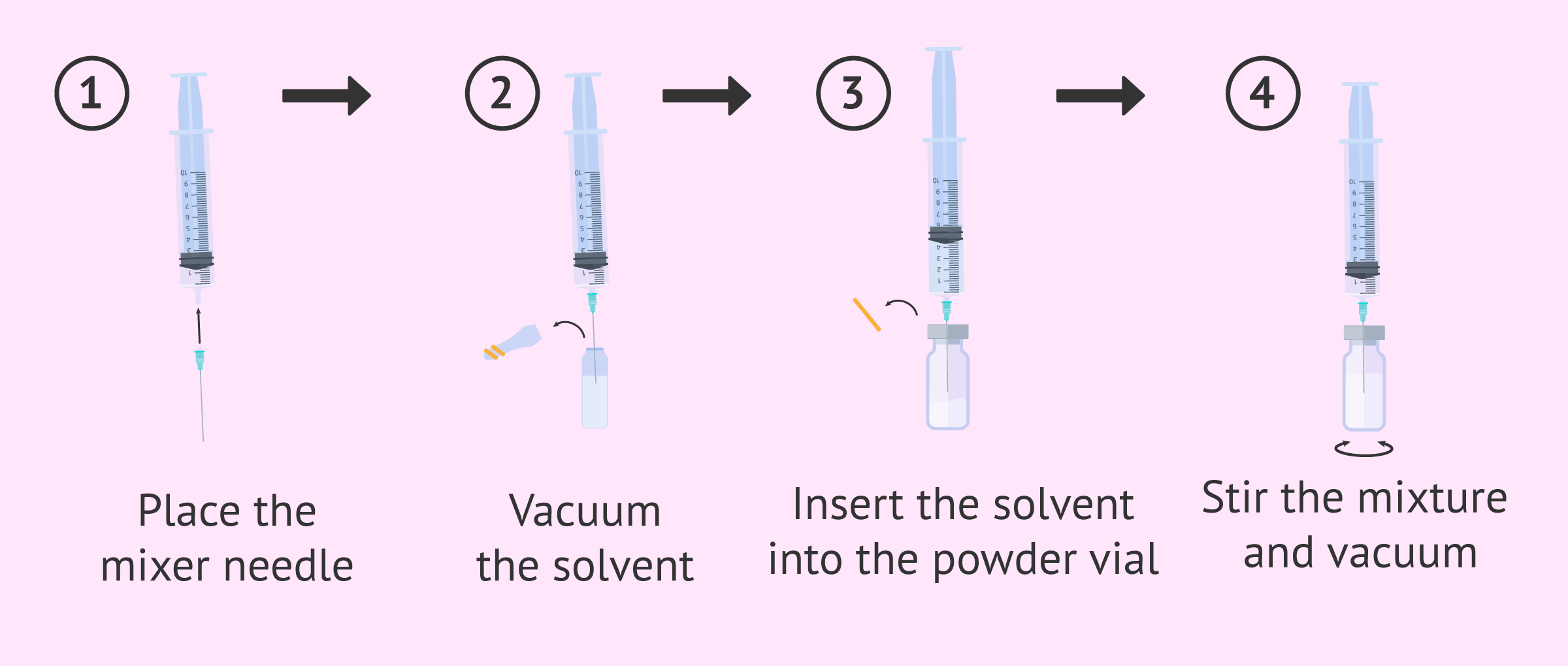
- Place the large (intramuscular) needle in the syringe and insert it into the ampoule of solvent.
- Retrieve the entire contents of the vial and slowly insert it into the powder vial, punching in the centre of the rubber stopper and reaching the end of the vial.
- In order to facilitate the dissolution of the powder, a gentle hand movement is made without removing the needle or releasing the plunger.
The next step will be to recover the dose that the gynecologist has prescribed and change the needle for the subcutaneous one. In order to remove any air in the syringe, hold the syringe with the needle upwards and tap it gently until air collects at the top. Then slowly press the plunger until all the air is removed.
Self-injection
Before self-injecting the drug, it is recommended to clean the area of the abdomen where the drug is to be administered with an alcohol-soaked gauze. It is also recommended to change the injection point of the dose every day, to protect the skin.
To administer the medication, pinch a little skin, insert the needle at a 45° to 90° angle and press the plunger slowly and steadily to the end. Then, it is recommended that you press a couple of seconds with a gauze over the area, massaging gently so that the drug disperses under the skin.
Another point to keep in mind is that the injection should be made immediately after preparation and at approximately the same time each day.
Contraindications
Tryptorelin is contraindicated in patients with hypersensitivity to GnRH, its analogues or any of its excipients.
It is also contraindicated in patients who are or may be pregnant and in breastfeeding periods. B
efore starting a treatment with Decapeptyl, it should be taken into account that prolonged treatments with GnRH agonists produce a reduction in bone mineral density.
Therefore, in patients with osteoporosis or risk factors for osteoporosis, it should be assessed whether the benefits of treatment outweigh the risk that it may pose. However, in most women, after therapy, bone density returns.
Possible adverse effects
Decapeptyl usually has no significant side effects. Most of the adverse reactions that occur are the result of changes in hormone levels, such as hot flashes, headaches, mood swings, decreased libido or sleep disturbances. Genital bleeding, vulvovaginal dryness and dyspareunia (pain with sexual intercourse) have also been described.
Another side effect that may occur is localized irritation at the injection site of the drug.
FAQs from users
I don't get my period after using three-month Decapeptyl. Can I be pregnant?
The probability of being pregnant is minimal, as decapeptyl is a medication that inhibits ovulation at a central level, making it virtually impossible for ovulation to occur.
The ideal thing would be to do an ultrasound scan to confirm that there is no pregnancy.
Can I store Decapeptyl in the fridge?
Decapeptyl should be kept at room temperature, not exceeding 25°C. If the dose used is less than that contained in a vial, the excess should be kept in the refrigerator. It is recommended that 5 minutes before it is administered, it should be taken out of the refrigerator to be tempered. It must also be protected from light until it is to be used.
Do I have to start the fertility treatment with Decapeptyl as soon as I'm on my period?
There are different protocols in which Decapeptyl is used. In those where you start at the beginning of the cycle, it is usually recommended that you start the treatment on day 2 of the cycle. That is, the day after having your period.
Suggested for you
Decapeptyl is used in the process of ovarian stimulation both to cause pituitary suppression and to induce ovulation. If you want to know the procedure in detail, you can access the following post: What is ovarian stimulation?
Community and Support
At inviTRA we work to make monthly and rigorous information accessible to everyone. If this article has helped you, consider supporting us so we can continue accompanying more people on their journey to parenthood.
References
FAQs from users: 'I don't get my period after using three-month Decapeptyl. Can I be pregnant?', 'Can I store Decapeptyl in the fridge?' and 'Do I have to start the fertility treatment with Decapeptyl as soon as I'm on my period?'.