The EndomeTRIO test consists of a very complete analysis of the state of the endometrium with the aim of increasing the chances of pregnancy during an in vitro fertilisation (IVF) treatment.
In reality, EndomeTRIO includes 3 endometrial tests to study the uterine condition at different levels and obtain a comprehensive diagnosis:
- ERA test
- EMMA test
- ALICE test
The main reasons for carrying out the EndomeTRIO test are recurrent miscarriages and repeated implantation failures.
Provided below is an index with the 10 points we are going to expand on in this article.
- 1.
- 2.
- 2.1.
- 2.2.
- 2.3.
- 3.
- 4.
- 5.
- 6.
- 6.1.
- 6.2.
- 6.3.
- 6.4.
- 7.
- 8.
- 9.
- 10.
What does endometrial factor mean?
The endometrium is the inner layer of the uterus where embryo implantation and the beginning of pregnancy takes place. For this purpose, the endometrium has the ability to regenerate itself with each menstrual cycle and become receptive during a woman's fertile days.
However, there are cases in which the endometrium may be altered and not have the ideal characteristics for implantation to occur. This would be what is known as endometrial factor infertility and it happens in 20% of cases.
For a successful fertility treatment, it is not only important to obtain good quality embryos with maximum implantation capacity, but it is also essential to have an optimal endometrium that allows implantation.
The causes of endometrial factor can be diverse, such as displacement of the implantation window, endometritis, refractory endometrium, and so on.
What is EndomeTRIO?
The EndomeTRIO test aims to analyze a woman's endometrial health in a very comprehensive way.
To do this, we need to do an endometrial biopsy from which 3 different tests are performed: ERA, EMMA and ALICE. Each of these tests provides relevant information on the condition of the endometrium.
We comment on each of these tests below.
ERA test
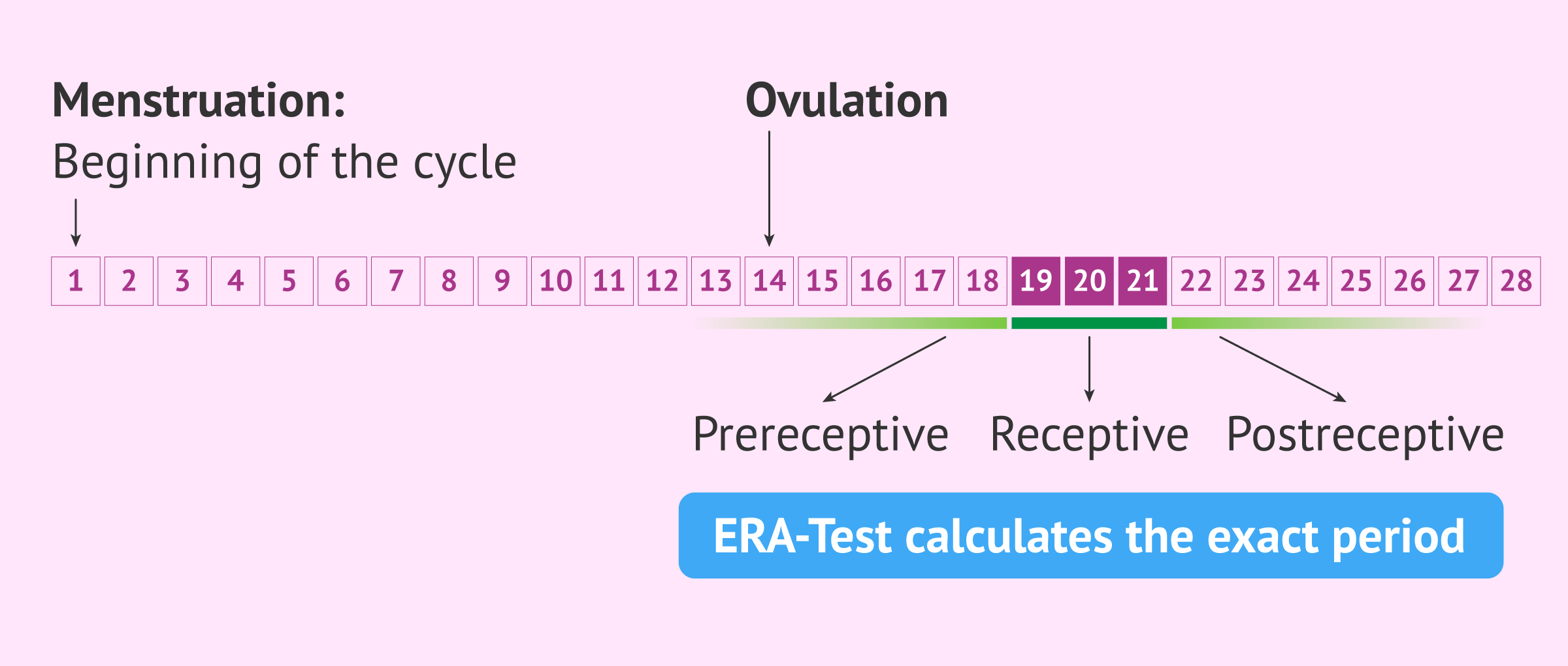
The ERA (Endometrial Receptivity Array) test is a molecular test that aims to identify the exact days of the implantation window and thus schedule the embryo transfer at the most suitable time.
To identify exactly which days of the cycle the endometrium is receptive, the expression of 248 genes is studied using the mass sequencing technique (NGS).
The result of the ERA test will indicate the state of the endometrium at the time of the biopsy: receptive, early receptive, late receptive, non receptive, etc.
If you are interested in learning more about this detailed studuy, you can continue reading about it in this article: Endometrial Receptivity Array (ERA): Procedure and Success Rates.
EMMA test
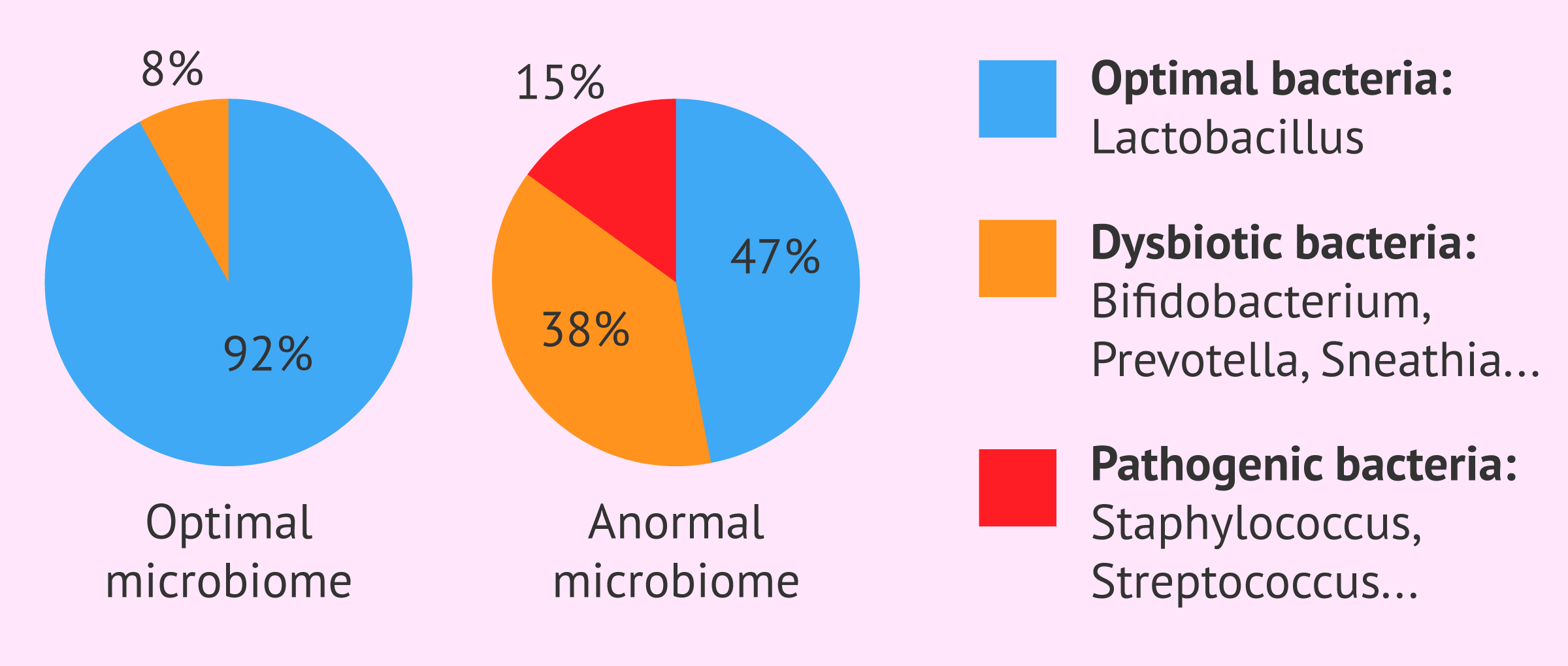
The EMMA test (Endometrial Microbiome Metagenomic Analysis) analyses the endometrial microbiome, i.e. the type and number of microorganisms in the uterine cavity, to determine whether it is optimal or not.
Recent studies have shown that pathogenic deviations in the levels of endometrial Lactobacilli and dysbiotic bacteria can lead to infertility, hence the importance of studying the microbial environment of the uterus.
The results of the EMMA test will include the percentage of Lactobacilli or other bacteria detected, as well as suggested antibiotic or probiotic treatment if necessary.
ALICE test
The ALICE (Analysis of Infectious Chronic Endometritis) test is a molecular test to diagnose chronic endometritis, which affects approximately 30% of infertile patients.
Chronic endometritis is a uterine infection caused by pathogenic bacteria that results in persistent inflammation of the endometrium.
The ALICE test identifies the pathogenic bacteria that cause chronic endometritis and how much of it is found in the endometrium. These bacteria are Enterococcus sp., Enterobacteriaceae (Escherichia and Klebsiella), Streptococcus sp., Staphylococcus sp., Mycoplasma sp., and Ureaplasma sp. ALICE also identifies other pathogenic bacteria associated with sexually transmitted diseases, such as Chlamydia and Neisseria.
Additionally, ALICE provides information on the most recommended antibiotic and/or probiotic treatment to cure endometritis.
What is the procedure for the EndomeTRIO test
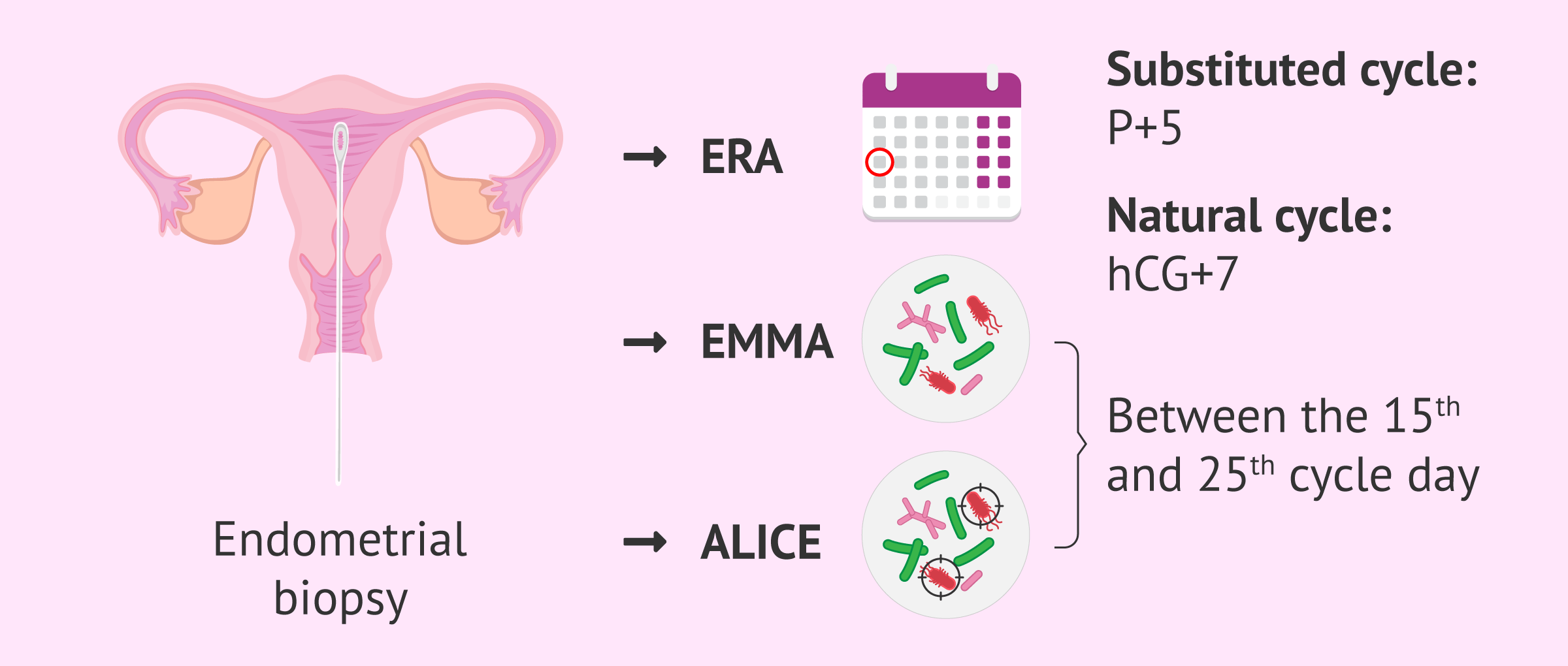
As we have already indicated, the first step to perform EndomeTRIO is to perform an endometrial biopsy on the patient and take a sample. The gynecologist will have to use a cannula and take the biopsy of the fundus of the uterus.
When considering the best day to do the endometrial biopsy, there are two options:
- EndomeTRIO with ERA test
- the endometrial biopsy should be taken under the same conditions under which the embryo transfer will subsequently be performed, i.e. in a substituted cycle or in a natural cycle. In the first case, the biopsy will be performed on day P+5 of progesterone administration, with day P+0 being taken on the first day of progesterone intake. In case of being in a natural cycle, the endometrial biopsy will be performed 7 days after the hCG injection, that is, on day hCG+7.
- EndomeTRIO without ERA test
- if only the EMMA and ALICE tests are performed, the endometrial biopsy can be taken between days 15 and 25 of the menstrual cycle.
In either case, the instructions and advice given by the specialist should be followed.
Indications of EndomeTRIO
Although any patient who is going to undergo assisted reproduction techniques can opt for EndomeTRIO, its main indications are the following:
- Implantation failures
- Repeated miscarriage
- Chronic Endometritis
- Endometriosis
- Hydrosalpinx
- Previous sexually transmitted diseases
As we have said, EndomeTRIO can also be an option for other patients who simply want to know the status of their endometrium, which will allow for a more personalized embryo transfer and an increase IVF success rates
Fertility treatments, like any other medical treatment, require that you trust the fertility specialists that will be by your side during your journey. Logically, each clinic has a different work methodology. Our Fertility Report will offer you a selection of recommended clinics, that is, fertility centers that have passed our rigorous selection process. Moreover, our system is capable of comparing the costs and conditions of each one so that you can make a well-informed decision.
Video about the EndomeTRIO test
Ana Fernández-Sanguino, gynaecologist at Fertility Madrid, talks to us in this video about the endomeTRIO test. As the doctor tells us:
The TRIO test studies three aspects of the endometrium.
FAQs from users
What is the EndomeTRIO test and what results does it offer?
The EndomeTRIO test is used to find out more about the patients' endometrium. It is a complete analysis of endometrial health that includes the following tests: ERA, EMMA and ALICE.
The ERA test analyses endometrial receptivity, the EMMA test analyses endometrial microbiota to increase reproductive success and the ALICE test detects the bacteria that cause chronic endometritis.
Numerous studies show the importance of a good endometrium as one of the fundamental keys to assisted reproduction treatment, as it is known that 20% of infertility is due to an endometrial factor.
Read more
What are the differences between the EMMA test and traditional microbial culture?
Microbial culture is a diagnostic method that consists of cultivating a biological sample in a plate with the optimal medium and conditions to promote the proliferation of microorganisms and to be able to identify them. In contrast, the EMMA test uses mass sequencing technology to identify genes that belong to microorganisms and not to the human species.
The advantage of the EMMA test is that it can identify bacteria that may not be detectable by culture or that are not directly cultivable.
How much do the endomeTRIO tests cost?
The endomeTRIO test includes three different tests to study the endometrium: ERA, EMMA and ALICE. The costs in the USA depend on your particular clinic, and are not usually covered by insurance, and are approximately as follows:
- ERA around $800
- EMMA approx $500
- ALICE approx $300
It is common for these tests to be ordered together and as a group should cost around $1000.
Does the endomeTRIO test hurt?
This test requires the performance of an endometrial biopsy and it is possible to feel some discomfort. This discomfort is similar to menstrual symptoms, so it is sometimes recommended to take a painkiller before the test is performed.
However, this discomfort is short-lived since the endometrial biopsy is very quick. Once completed, the patient should not feel any pain.
Recommended reading
If you want to know more about the endometrium and its role in the menstrual cycle, you can continue reading here: What’s the Role of the Endometrium? Function & Thickness.
We have said that the EndomeTRIO test is useful to optimize an IVF treatment and to achieve a better success rate. If you want to learn more about the process, you can access the following post: What is in vitro Fertilization? Process, Cost & Success Rates
Finally, EndomeTRIO will provide information on the best time to perform the embryo transfer and achieve a positive result. Learn more about the embryo transfer here: IVF Embryo transfer procedure - definition, process & tips
Community and Support
At inviTRA we work to make monthly and rigorous information accessible to everyone. If this article has helped you, consider supporting us so we can continue accompanying more people on their journey to parenthood.
References
Cicinelli E, Matteo M, Tinelli R, Lepera A, Alfonso R, Indraccolo U, Marocchella S, Greco P, Resta L. Prevalence of chronic endometritis in repeated unexplained implantation failure and the IVF success rate after antibiotic therapy. Hum Reprod, 2015; 30(2):323-30 (view)
Díaz-Gimeno P, Horcajadas JA, Martínez-Conejero JA, Esteban FJ, Alamá P, Pellicer A, Simón C. A genomic diagnostic tool for human endometrial Receptivity based on the transcriptomic signature. Fertil Steril. 2011; 95(1):50-60, 60.e1-15 (view)
Mahajan N. Endometrial receptivity array: Clinical application. J Hum Reprod Sci. 2015; 8(3):121-9 (view)
Moreno I, Cicinelli E, Garcia-Grau I, Gonzalez M, Bau D, Vilella F, De Ziegler D, Resta L, Valbuena D, Simon C. The diagnosis of chronic endometritis in infertile asymptomatic women: a comparative study of histology, microbial cultures, hysteroscopy, and molecular microbiology. Am J Obstet Gynecol. 2018; 218(6):602.e1-602.e16 (view)
Moreno I, Codoñer FM, Vilella F, Valbuena D, Martinez-Blanch JF, Jimenez-Almazan J, Alonso R, Alama P, Remohi J, Pellicer A, Ramon D, Simon C. Evidence that the endometrial microbiota has an effect on implantation success or failure. Am J Obstet Gynecol. 2016; 215:684-703
Ruiz-Alonso M, Blesa D, Díaz-Gimeno P, Gómez E, Fernández-Sánchez M, Carranza F, Carrera J, Vilella F, Pellicer A, Simón C. The endometrial Receptivity array for diagnosis and personalized embryo transfer as a treatment for patients with repeated implantation failure. Fertil Steril. 2013; 100(3):818-24 (view)
FAQs from users: 'What is the EndomeTRIO test and what results does it offer?', 'What are the differences between the EMMA test and traditional microbial culture?', 'Why is it called EndomeTRIO?', 'How much do the endomeTRIO tests cost?', 'Is it possible to do only two of the three analyses that make up the EndomeTRIO test?', 'Is the EndomeTRIO test indicated in all patients?', 'Does the endomeTRIO test hurt?', 'What are the steps to perform the EndomeTRIO test?' and 'What are the possible results of the EndomeTRIO test?'.
Authors and contributors

More information about Michelle Lorraine Embleton







I had this test after 5 failed transfers and it came out that I had endometritis. I highly recommend it because despite being expensive, it is worth getting tested rather than going blindly through the treatment. Now I am preparing my endometrial lining for the next transfer and I hope it will be the last one!