Ovitrelle, also called Ovidrel in the U.S., is a drug that contains the hormone recombinant hCG (r-hCG) and is used in the vast majority of assisted reproduction treatments.
It consists of a single injection that manages to trigger the final follicular maturation after controlled ovarian stimulation, both in artificial insemination (AI) and in vitro fertilization (IVF).
Ovitrelle is expensive, but it is very effective, which has allowed the optimization of ART cycles and many women to achieve pregnancy.
Provided below is an index with the 10 points we are going to expand on in this article.
- 1.
- 2.
- 2.1.
- 2.2.
- 3.
- 3.1.
- 3.2.
- 4.
- 5.
- 6.
- 6.1.
- 6.2.
- 6.3.
- 6.4.
- 6.5.
- 6.6.
- 6.7.
- 7.
- 8.
- 9.
- 10.
Composition of Ovitrelle
The active principle of Ovitrelle is choriogonadotropin alpha, a recombinant hCG hormone obtained in the laboratory thanks to genetic engineering techniques, so it is artificial in origin. As for the formats in which Ovitrelle is found, we find these two:
- Ovitrelle in pre-filled syringe
- Ovitrelle in pre-filled syringe consists of a container with 1 pre-filled syringe containing 250 µg of choriogonadotropin alpha in 0.5 ml of solution for injection, which is equivalent to approximately 6,500 IU.
- Ovitrelle in pre-filled syringe
- is supplied in a container with 1 pre-filled pen containing 250 µg of alpha choriogonadotropin in 0.5 ml of solution for injection, equivalent to approximately 6,500 IU.
In addition to its active ingredient, choriogonadotropin alpha, other excipients of the Ovitrelle drug are mannitol, methionine, disodium hydrogen phosphate dihydrate, sodium dihydrogen phosphate monohydrate, poloxamer 188, phosphoric acid, sodium hydroxide and water for injection. Currently, the pre-filled pen is the most used format.
Mechanism of action
Since women undergoing fertility treatment are previously hormonally suppressed, i.e. they do not produce endogenous pituitary hormones and their cycle is exogenously controlled by hormonal drugs, it is also necessary to administer an injection of the hormone hCG in order to trigger ovulation.
r-hCG (recombinant human chorionic gonadotropin) acts as a peak substitute for LH (luteinizing hormone), which would trigger ovulation during a normal menstrual cycle.
Specifically, Ovitrelle performs the following actions on the woman's body:
- It promotes the resumption of oocyte meiosis and thus induces the final maturation of the follicles that have developed in the ovarian stimulation.
- It allows ovulation (release of the mature egg from the ovary) in artificial insemination or programmed coitus cycles so that fertilization can take place.
- It triggers the formation of the corpus luteum in the ovary, which will secrete estradiol and the progesterone necessary for the endometrium to become receptive and for embryo implantation to occur.
Specifically, Ovitrelle promotes the final maturation of the eggs and their release approximately 36-48 hours after administration. Therefore, it is very important to schedule the artificial insemination or, in the case of IVF, the follicular puncture about 34 hours after injecting Ovitrelle.
It is essential to respect the injection times set by the doctor. Otherwise, this drug could negatively interfere with the final outcome of the reproductive treatment.
Indications
For all that has been said so far, Ovitrelle is indicated in the following situations:
- Women with anovulation or oligovulation, that is, who are not able to ovulate or who produce very few eggs. One of the most frequent cases of anovulation is polycystic ovary syndrome (PCOS).
- Women undergoing an artificial insemination cycle, to induce ovulation and introduce the capacitated sperm of the partner or of an anonymous donor.
- Women undergoing an IVF cycle, to trigger follicular maturation and to schedule egg retrieval before ovulation occurs. Several mature eggs can then be obtained for fertilization in the laboratory.
- Women undergoing a natural cycle for the transfer of frozen embryos or embryos from egg donation. In these cases, Ovitrelle is used to force ovulation and start with the supply of progesterone.
It should be noted that Ovitrelle can only be used under indication and prescription.
Assisted procreation, as any other medical treatment, requires that you rely on the professionalism of the doctors and staff of the clinic you choose. Obviously, each clinic is different. Get now your Fertility Report, which will select several clinics for you out of the pool of clinics that meet our strict quality criteria. Moreover, it will offer you a comparison between the fees and conditions each clinic offers in order for you to make a well informed choice.
Contraindications
On the other hand, the use of Ovitrelle is not recommended in some patients because of the risks it might possess to their health. These are the situations in which Ovitrelle is contraindicated:
- Allergy to the active ingredient or to some of its excipients.
- Tumors in the ovaries, uterus or breasts, as well as tumors in the hypothalamus or pituitary gland.
- Vaginal bleeding with no known cause.
- Presence of cysts in the ovaries not caused by PCOS.
- History of ectopic pregnancy in the last 3 months.
- Active thromboembolic problems.
In addition, although not a contraindication, it is not recommended to use Ovitrelle in situations where an effective response is not expected or when the woman has circumstances that prevent her from carrying a normal pregnancy.
Some examples of these situations are primary ovarian failure, malformations in the sexual organs incompatible with pregnancy, or fibroid tumors of the uterus incompatible with pregnancy.
Instructions of use
In both formats of Ovitrelle, the drug is administered subcutaneously, in other words, it is injected under the skin.
However, in this section, we will refer to the mode of administration of Ovitrelle with the preloaded pen, as this is the format most used today.
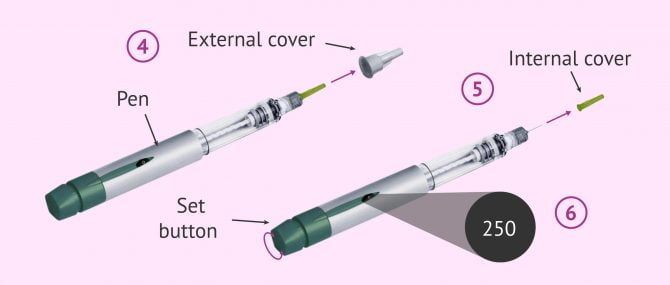
Preparation of the pen
Firstly, it must be clear that each Ovitrelle pre-filled pen is for single use only. For its preparation, the following instructions must be adhered to:
- Remove the cap from the pen.
- Prepare the needle for injection: hold the outer needle sleeve firmly and remove the locking tab.
- Screw the threaded end of the preloaded pen to the external needle sleeve until the needle is well attached.
- Remove the outer cover of the needle by gently pulling it out.
- Hold the preloaded pen with the needle pointing upwards and carefully remove the green internal protector.
- Set dose to 250: Gently turn the dose setting knob until the dose indicator shows '250'.
How to inject Ovitrelle in a pre-filled pen?
Once the Ovitrelle pen has been prepared, the patient should proceed with her injection as instructed by her doctor or nurse. In general, the following steps are taken:
- Clean the injection area with an alcohol swab.
- Slightly pinch the skin in the abdominal area where the injection is to be made.
- Slowly insert the entire needle into the skin.
- Press the dose setting button fully and hold it down for about 5 seconds to make sure you have injected all the drug (the dose number on the indicator will return to '0').
- Remove the needle from the skin by keeping the dose setting button pressed.
Finally, it is also very important to dispose of the injection needle and the used pen in the correct way, as instructed by your pharmacist.
Risks and side effects
The administration of Ovitrelle may produce certain symptoms and/or unwanted side effects. The most common ones are the following:
- Headache
- Fatigue
- Local reactions in the injection area
- Swelling or abdominal pain
- Gastrointestinal disorders, such as diarrhea or nausea
- Breast pain
- Mild or moderate ovarian hyperstimulation syndrome (OHSS)
Although less frequent, there are other situations in which the use of Ovitrelle can cause more serious effects such as psychiatric disorders, allergic skin reactions, rashes, severe OHSS, etc.
Ovitrelle and pregnancy test
On multiple occasions, we have commented that taking a pregnancy test early after a fertility treatment can result in a false positive.
This is due to the effect of Ovitrelle, as this drug contains the recombinant hCG hormone, which can be detected by pregnancy tests.
A pregnancy test measures the level of the hormone beta-hCG, known as the pregnancy hormone because it is only found in a woman's body when implantation of the embryo in the uterus takes place and gestation begins.
Therefore, it can happen that the pregnancy test detects hCG from Ovitrelle instead of the beta-hCG released by the implanted embryo, resulting in a positive result that is actually wrong.
As a consequence of this and to avoid obtaining false positives, it is important that the woman takes the pregnancy test at least 14 days after the follicular puncture or artificial insemination as the case may be.
The woman's body must be given enough time to eliminate the remains of Ovitrelle in the blood after administration.
You can find out more about this in the following article: When to do a pregnancy test?
FAQs from users
Is Gonal the same as Ovitrelle?
No. Gonal and Ovitrelle are fertility drugs with different functions and different mechanisms of action.
Gonal consists of recombinant FSH (follitropin alpha) and is used to promote the growth of follicles during ovarian stimulation. Ovitrelle is a drug that contains the recombinant hCG hormone (r-hCG) and is used in the vast majority of assisted reproduction treatments. It involves a single injection that induces the final follicular maturation after controlled ovarian stimulation, both in artificial insemination (AI) and in vitro fertilization (IVF).
Read more
How and when should Ovitrelle be administered?
Ovitrelle should always be administered subcutaneously and under medical supervision. The administration takes place once the optimal follicle size is reached to trigger ovulation, which coincides with the middle of the menstrual cycle.
How many days is Ovitrelle detectable in the blood?
Ovitrelle is a hormone called chorionic gonadotropin. It is administered to induce ovulation in those women who do not ovulate spontaneously or after ovarian stimulation as part of an assisted reproduction treatment.
Depending on the bioavailability of the drug, its half-life is 30 hours, after which most of it will be eliminated from the body. However, there may be interference in blood and urine tests up to 10 days after administration, which may lead to false-positive pregnancy tests. It is important that patients are warned to always take a pregnancy test at least 11 days after Ovitrelle has been administered.
Is it normal to administer Ovitrelle after embryo transfer?
Although ovitrelle is especially indicated to promote ovulation and is therefore administered just before artificial insemination or ovarian puncture, some studies show certain advantages of administering this drug after the transfer of the embryos in IVF cycles.
In any case, it is something experimental for which no significant results have been obtained yet.
Does menstruation start earlier with Ovitrelle?
Ovitrelle is used to promote the final maturation of the eggs and ovulation, but it does not necessarily have to forward the start of menstruation. You will be on your period approximately two weeks after ovulation caused by Ovitrelle unless pregnancy has occurred, in which case there will be no menstruation.
Does alcohol consumption negatively influence the effect of Ovitrelle?
No, alcohol does not have to prevent Ovitrelle from working. However, the consumption of alcohol is totally inadvisable when you are trying to get pregnant, whether naturally or through fertility treatment.
How much does Ovitrelle cost?
Depending on the pharmacy, the costs of ovitrelle are $390 per unit.
Suggested for you
We have said that Ovitrelle is used in the vast majority of assisted reproduction cycles during the ovarian stimulation phase. If you want to know what this is and what other medications are used, you are welcome to visit the following post: What is ovulation induction?
If you are also interested in knowing which are the sex hormones that participate in all the reproductive functions of men and women, we advise you to continue reading here: male and female sex hormones.
Community and Support
At inviTRA we work to make monthly and rigorous information accessible to everyone. If this article has helped you, consider supporting us so we can continue accompanying more people on their journey to parenthood.
References
FAQs from users: 'Is Gonal the same as Ovitrelle?', 'How and when should Ovitrelle be administered?', 'How many days is Ovitrelle detectable in the blood?', 'Is it normal to administer Ovitrelle after embryo transfer?', 'Does menstruation start earlier with Ovitrelle?', 'Does alcohol consumption negatively influence the effect of Ovitrelle?' and 'How much does Ovitrelle cost?'.
Authors and contributors